Int. J. Dev. Biol. 69: 161 - 172 (2025)
Sialylated glycoproteins and sialyltransferases involved in mesoderm-derived organ formation during embryogenesis
Open Access | Review | Published: 6 April 2026
Abstract
Most major organs, like muscles, bones, vessels and kidneys, develop from the mesoderm, one of three germ cell layers in triploblastic organisms. Sialic acids significantly affect embryonic development by regulating cell division, migration and death through signaling pathways and cell adhesion, which support morphogenesis. Loss of early biosynthetic enzymes reduces embryonic viability and leads to complex phenotypes, while the loss of terminal enzymes primarily results in tissue-specific defects in mesoderm-derived organs. Key sialylated glycoproteins involved in the developmental processes of mesoderm and mesoderm-derived organs have been identified across various species as major effectors. These enzymes and glycoproteins are of significant interest and are discussed in the present review.
Keywords
sialic acids, sialyltransferases, mesoderm, embryonic development, organogenesis
Introduction
A dense complex glycocalyx, composed of numerous glycoconjugates including glycoproteins and glycolipids, coats cell surfaces. Sialic acids represent the predominant terminal sugar residues of glycoconjugates found on most cells’ surfaces. Sialic acids consist of a group of nine-carbon amino sugars that are positioned at the nonreducing terminal ends of N- or O-glycoproteins and glycolipids, forming the so-called sialome (Cohen and Varki, 2010; Sato and Kitajima, 2021; Varki, 2017). Their high structural diversity has been shown to participate in various cellular functions during different stages of embryonic development.
Following fertilization and cleavage, the mesoderm, ectoderm, and endoderm form and rearrange during gastrulation, involving cell movement, interactions, and regulation of adhesion factors via ligand-receptor signals involving Sia. Additionally, epithelial-mesenchymal transition (EMT) occurs, alongside the regulation of sialylation. Most major organs develop afterwards during early and late organogenesis from the mesoderm, where sialylation influences the formation of structures, including the skeleton (cartilage and bones), muscles (skeletal and cardiac), kidneys (pronephros and metanephros), the blood and lymphatic vasculature (endothelial and lymphatic vessels). Although the function of sialylation in ectoderm and nervous system development has been thoroughly investigated (see Schnaar et al., 2014; Sato and Kitajima, 2021), its role in the formation of endoderm and particularly in mesoderm and mesoderm-derived organs has received less attention in current reviews.
This review discusses the roles of biosynthetic enzymes and sialyltransferases in the formation of the mesoderm and its derived organs, as well as potential effectors regulated by Sia. Disruption of early cytosolic enzymes in the sialic acid biosynthesis pathway, such as the glucosamine UDP-GlcNAc-2-epimerase/N-acetylmannosamine kinase (encoded by the GNE gene), can result in death or improper development of both ectodermal derivatives and mesoderm-derived organs (Varki and Schauer, 2009). Knockouts, knockdowns, and in vitro studies have identified key enzymes for sialic acid biosynthesis in mesoderm-derived organs. However, many sialylated effectors and sialyltransferases remain unclear due to their complex and potentially compensatory roles. These components may be valuable for understanding and treating diseases, but further research is needed to define their functions and regulatory networks.
Sialylated glycoconjugates are characterized by a high structural diversity
Sialic acids are derived from N-Acetylneuraminic acid (Neu5Ac) and 2-keto-3-deoxynononic acid (Kdn). Neu5Ac and 5-N-glycolylneuraminic acid (Neu5Gc). The two most abundant are Neu5Ac and 5-N-glycolylneuraminic acid (Neu5Gc) are the most abundant forms, followed by Kdn and neuraminic acid (Neu), which is almost not present in nature (Schauer, 1982). The diversity of sialoglycoconjugates arises from the combination of sialic acid structures, substitutions, degree of polymerization (DP), linkages, and the nature of the related glycans. Hydroxyl or amino groups can be substituted with acetyl, glycolyl, methyl, lactyl, phosphate, or sulfate groups, generating more than 50 structurally different derivatives (Schauer and Kamerling, 2018). Notably, acetyl and sulfate groups play a crucial role in vertebrate embryos (Ertunc et al., 2022; Varki and Schauer, 2009; Varki et al., 1991), Fig. 1A.
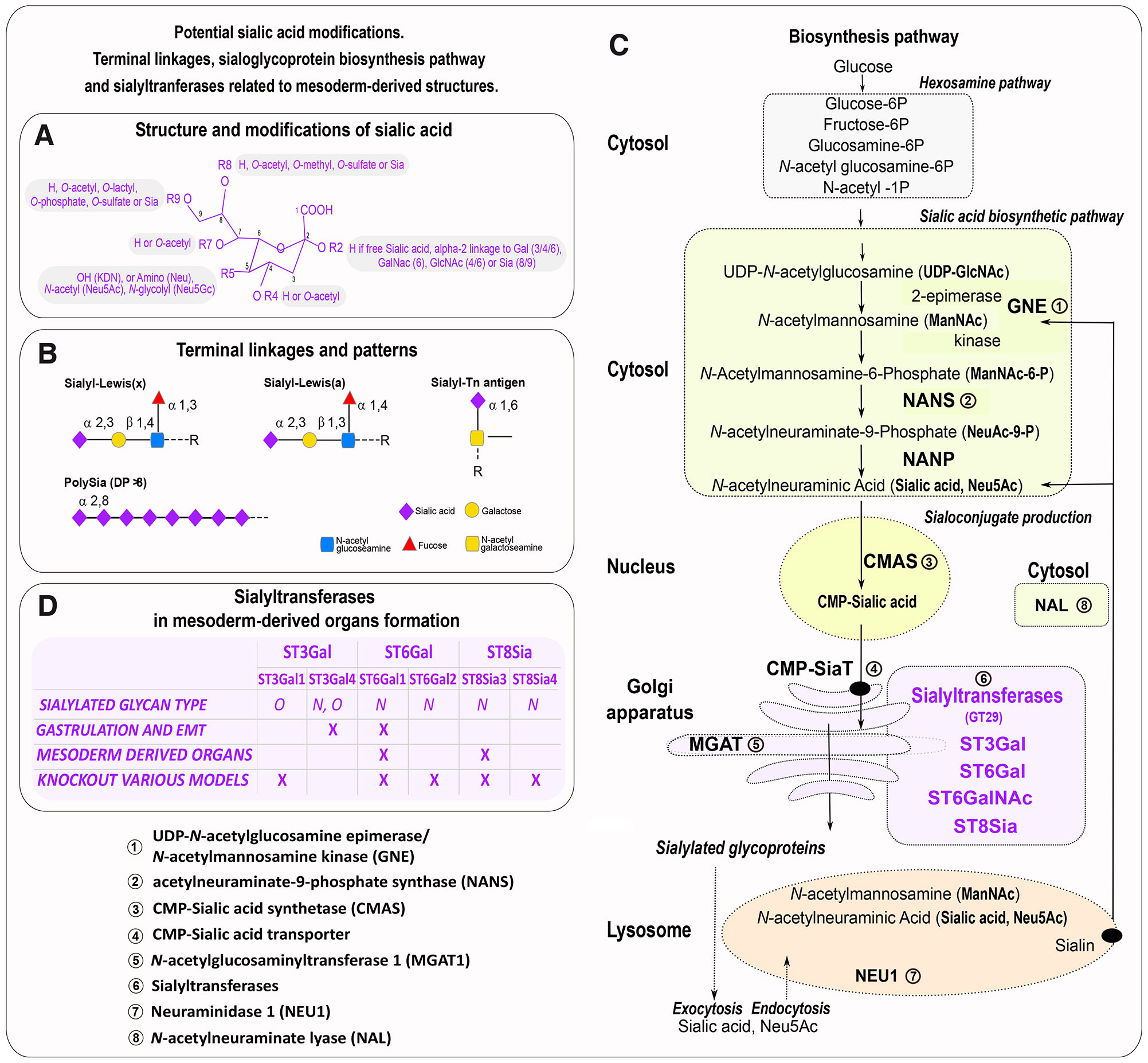
Fig. 1. Sialoglycoprotein biosynthesis pathway, sialyltransferases involved in mesoderm-derived organs, and sialic acid structure.
(A) Sialic acid structure and modifications. (B) Terminal linkage and patterns found during embryonic development in mesoderm-derived organs. (C) Sialic acid biosynthesis pathway and associated enzymes detected in mesoderm-derivatives; the enzymes involved in mesodermal organs formation are in bold. (D) Sialyltransferases involved in gastrulation, epithelial to mesenchymal transition (EMT), and mesoderm-derived organs formation in various species. The color refers to the intracellular location of enzymes as follows: cytosol (green), nucleus (yellow), Golgi (orange), and lysosome (pink). Sialyltransferases are purple.
Another source of diversity is the different glycosidic α-linkages that connect sialic acid moieties to other glycan units. These linkages vary between α2,3 and α2,6 linkages to galactose-containing glycans, as well as in α2,6 linkages to N-acetylgalactosamine (GalNAc) and N-acetylglucosamine (GlcNAc) residues. Distinct α2,8 linkages create sialic acid chains on certain glycoproteins, which can vary in their degree of polymerization (DP). Polysialic acid (PSA), in particular, is defined by having a DP greater than 8 (Sato and Kitajima, 2013; Troy, 1992), Fig. 1B. Finally, sialoglycoconjugates can assemble into cell surface micro-domains relaying biological signals (Sato and Kitajima, 2013; Troy, 1992).
Sialic acids are present in Metazoa such as Echinoderms (Miyata et al., 2006), Hemichordata, Cephalochorda, Vertebrata, Platyhelminthes, Cephalopoda and Crustacea. Additionally, they are reported in viruses, bacteria, protozoa, and fungi (Ghosh, 2020). While sialylation occurs in Drosophila embryos (Aoki et al., 2007; Frappaolo et al., 2017), evidence of sialylation in other insects is controversial, despite the detection of sialyltransferase homologs in their genome sequences (Kajiura et al., 2015; Koles et al., 2004; Paschinger and Wilson, 2019). C. elegans glycans lack sialic acids (Bacic et al., 1990). In addition, Neu5Gc is absent in humans and monotremes (including the platypus) and occurs only rarely in Sauropsids (birds and reptiles), (Altman and Gagneux, 2019; Chen and Varki, 2010). Deaminated neuraminic acid, or Kdn, is found abundantly in lower vertebrates and bacteria (Ghosh, 2020).
Sialic acid biosynthesis, a multistep pathway in embryos
The de novo biosynthesis of sialic acids originates from glucose metabolism and occurs through a series of steps located in different cellular compartments. Enzymes involved in the modification of sialic acids and sialyltransferases, which are important for the formation of mesoderm and mesoderm-derived organs, are highlighted in Fig. 1C.
The process begins in the cytosol, by epimerization and phosphorylation of UDP-GlcNAc (uridine diphosphate N-acetylglucosamine) into ManNAc-6-P (N-acetyl-mannosamine 6-phosphate) and UDP (uridine diphosphate) by the bifunctional enzyme UDP-GlcNAc-2-epimerase/N-acetylmannosamine kinase (encoded by the GNE/MNK gene). ManNAc-6-P is then condensed with phosphoenolpyruvate (PEP) by the enzyme Neu5Ac-9-P synthase (NANS) to produce N-acetylneuraminate 9-phosphate (Neu5Ac-9-P). This compound is subsequently dephosphorylated by N-acetylneuraminate-9-phosphatase (NANP) to generate Neu5Ac.
In the nucleus, CMP-Sia (cytidine monophosphate sialic acid) is catalyzed by the cytidine monophosphate N-acetylneuraminic acid synthetase (CMAS). The activated CMP-Sia is then transported within the Golgi apparatus, where sialylation occurs (Lepers et al., 1989; Lepers et al., 1990) using a CMP-Sia Transporter (CMP-SiaT)/ SLC35A1, a nucleotide sugar transporter. Glycosidic linkages are formed by specific α2,3-, α2,6- or α2,8-sialyltransferases from four families of sialyltransferases ST3Gal, ST6Gal, ST6GalNAc and ST8Sia (Harduin-Lepers et al., 2005; Chang et al., 2009; Teppa et al., 2016; Harduin-Lepers, 2023). Sialyltransferases detected in mesoderm and mesoderm-derived organs are shown in Fig. 1D. These enzymes belong to the glycosyltransferase family 29 (GT29), as documented in the Carbohydrate Active Enzymes database (CAZy, http://www.cazy.org) (Lombard et al., 2014).
O-acetylation and O-sulfation of sialic acids occur in the Golgi. The 9-O-Acetylation of sialic acids (Sia9Ac) arises at the CMP-Sia level using acetyl-coenzyme A (AcCoA) as a donor. It occurs in conjunction with Golgi-resident sialyltransferases. 8-O-sulfation of sialic acids employs O-sulfotransferases Wscd1 and Wscd2 after CMP-Sia transport into the Golgi lumen, sialyltransferases, and 3′-phosphoadenosine 5′-phosphosulfate (PAPS).
Other glycosyltransferases from the α-1,6-mannosylglycoprotein 6-β-N-acetylglucosaminyltransferase (MGAT) family and hexosyltransferases found in the Golgi apparatus, that generate branched glycans composed of N-acetyllactosamine (LacNAc) are ending with Sia, and involved in mesoderm and mesoderm-derivatives formation. The MGAT1 gene encodes N-acetylglucosaminyltransferase I (GlcNAc-TI), which initiates complex and hybrid N-glycan synthesis (Schachter, 2000). GlcNAc-TI allows mature N-glycoproteins to carry oligomannosyl with branched antennae that contain Sia.
Sialyl Lewisx (sLex) also known as cluster of differentiation 15 (CD15) or stage-specific embryonic antigen 1 (SSEA-1), is a sialic acid tetrasaccharide carbohydrate attached to O-glycans, and composed by fucose and N-acetyllactosamine (5-acetylneuraminyl-(2-3)-galactosyl-(1-4)-(fucopyranosyl-(1-3))-N-acetylglucosamine, Neu5Acα2-3Galβ1-4[Fucα1-3]GlcNAcβ). Another tetrasaccharide Sialyl-Lewisa (sLea), sialylated by an α2,3 linkage, differs in its linkages: it is a fucose group linked to N-acetylglucosamine by an α1,4 linkage instead of α1,3 linkage for sLex, while the galactose is linked to N-acetylglucosamine by a β1,3 linkage for sLea while in sLex it is linked by an β1,4 linkage. The biosynthesis of sLex in humans involves fucosyltransferases (CAzy GT10), notably FUT6 and FUT7 the main enzymes, FUT5 and FUT4 to a lesser extent, and FUT9 with a broad substrate specificity. FUT3 synthesizes sLea through an α1,4 linkage addition of fucose moieties, whereas FUT5 only performs it to a lesser extent. Fucosyltransferase activity occurs after the α2,3 linkage of sialic acids by various ST3Gal enzymes (Carvalho et al., 2010; Mondal et al., 2018). Tissues of mesodermal origin also express the Sialyl Tn antigen, an α2,6-sialylated N-acetylgalactosamine (GalNAc) linked by a glycosidic bond to serine or threonine. Terminal linkages and sialylated motifs found in mesoderm-derived organs are depicted in Fig. 1D.
Free sialic acids can be recycled following cleavage by neuraminidases/sialidases (NEU) which have distinct cell expression patterns. NEU1 is located in the lysosomes of Metazoa, where it hydrolyses exogenous sialoglycoconjugates. Four human NEU enzymes, classified in the glycoside hydrolase family GH33 according to the CAZy classification, have been described with specific expression patterns (Monti et al., 2010). Neu5Ac is then transported to the cytosol via a sialin/SLC17A5 transporter for the synthesis of CMPNeu5Ac. Cytosolic sialic acid catabolism occurs with N-acetylneuraminate lyase (NAL) acting on Neu5Ac to produce ManNAc (N-acetyl-mannosamine) and pyruvate (Schauer et al., 1999). ManNAc, can be converted into N-acetylglucosamine (GlcNAc) by N-acetylglucosamine 2-epimerase (encoded by the GNE gene), and subsequently phosphorylated to GlcNAc-6-phosphate, allowing it to reenter the hexosamine biosynthesis pathway (Varki et al., 2015). The availability and synthesis of sialic acids influence the activity of sialyltransferases (Bork et al., 2017). Despite this, the regulation of Sia, its metabolites, lysosomal recycling, and catabolism remain unclear.
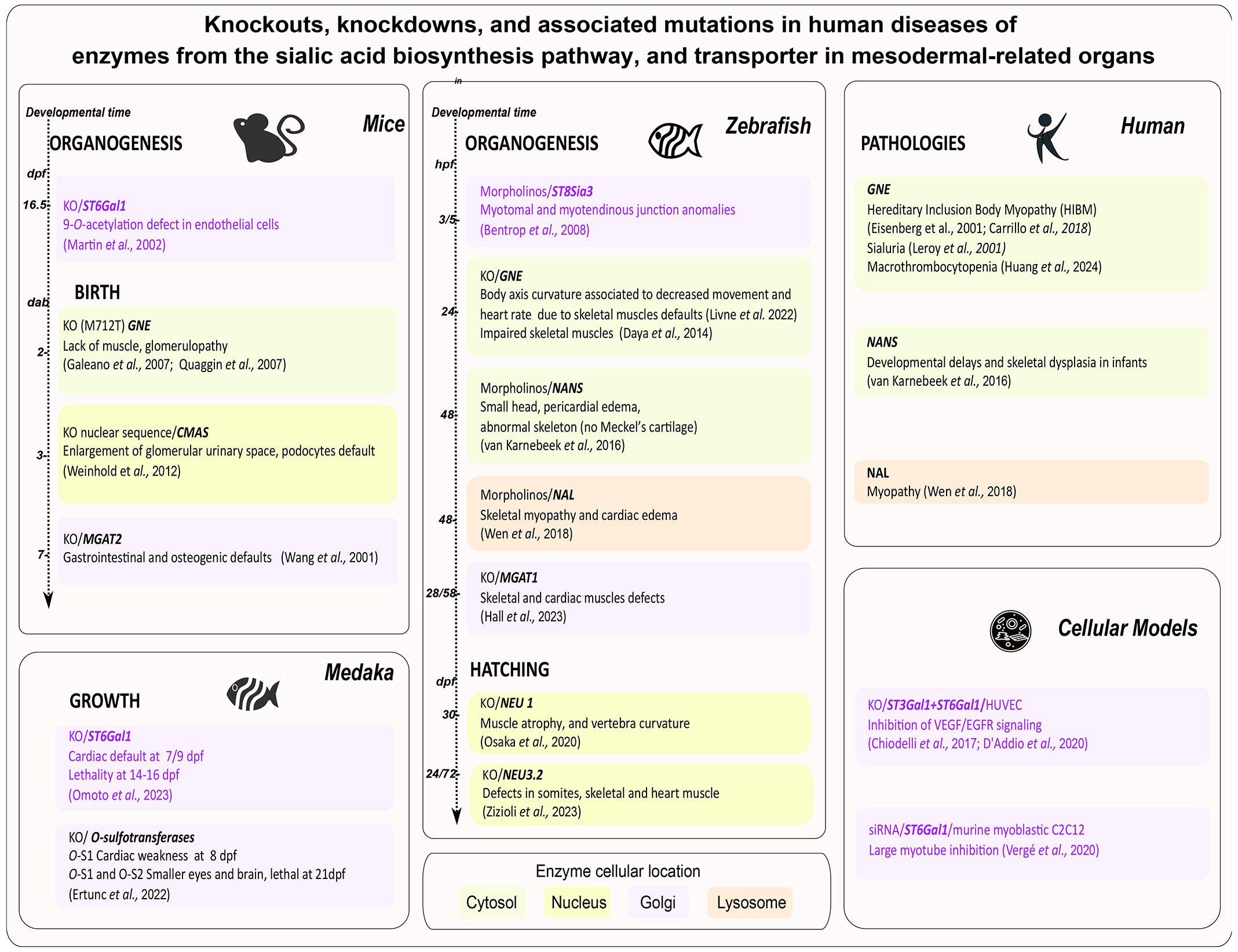
Enzymes from the sialic acid biosynthesis pathway linked to mesoderm-derived organs in different species, as evidenced by knockout, knockdown, or related human diseases, are discussed below and summarized in Fig. 2.
Fig. 2. Enzymes from the sialic acid biosynthesis pathway or transporter shown to be involved in the formation of mesoderm-derived organs and associated mutations in human diseases.
For mice and zebrafish, the time of the phenotype's appearance within the developmental program is chronological. For humans, mutations generating congenital diseases are mentioned. The background colors refer to the cellular location of mutated gene products: cytosol (green), nucleus (yellow), Golgi (orange), and lysosome (pink). Sialyltransferases are purple. Abbreviations: Days after birth (dab), days post fertilization (dpf), hours post fertilization (hpf), knockout (KO), knockdown (morpholinos).
Properties of sialic acid in cellular functions
Throughout development, various molecular effectors, including growth factors, receptors, channels, and adhesion molecules, orchestrate a spatiotemporal coordinated decisional program to regulate cellular division, migration, and subsequent morphological changes. Sialic acids have the potential to modulate intracellular signaling and cellular interactions, acting as “fine-tuners” of these processes, due to their negative charge and hydrophilicity, which make them anti-adhesive glycotopes. For instance, sialic acids on endothelial cell surfaces generate repelling electrostatic fields, enabling cell separation and central lumen formation in blood vessels (Abeln et al., 2019). The negative charge of sialic acids on cadherin enhances membrane repulsion of adjacent cells, thereby regulating cell adhesion (Born and Palinski, 1985; D’Addio et al., 2020; Varki and Gagneux, 2012).
Sialylated glycoproteins deliver external stimuli to cells, relay signals between cells, and facilitate communication during mesoderm formation at gastrulation and mesoderm-derived organ development. Sialic acids regulate receptor and channel activity by altering electrostatic interactions, either inhibiting or activating specific molecules: either negatively through negative repulsive charges of sialic acids between two opposite molecules such as for the Neural adhesion molecules (NCAM) (Galuska et al., 2010), or positively by an attractive force between the negatively charged sialylated vascular endothelial growth factor-A (VEGF) and positively charged amino acids to reinforce the activation of VEGFR2 receptor (Chiodelli et al., 2017). Sialic acids silence the hyaluronan receptor LYVE-1 in the lymphatic endothelium (Nightingale et al., 2009), impact receptor oligomerization as evidenced by the effect of α2,6-Sia on endothelial cell adhesion molecule (PECAM1/CD31) to modulate cell adhesion, and play a role in angiogenesis (Kitazume et al., 2010). Sialic acids and PSA also directly bind small cationic molecules, such as growth factors like fibroblast growth factor (FGF2) (Ono et al., 2012; Sato and Kitajima, 2013; Zhang et al., 2004), thus influencing their signaling at gastrulation or during endothelial vessels formation. Furthermore, polysialic acids (PSA) enhance FGFR signaling and cell migration (Li et al., 2011). Interestingly, long chains of sialic acids also control channel activity. PSA regulate the α subunit of the voltage dependent sodium channel influencing the rapidity of closure and opening In rat embryonic myocardium cells (4 to 5 days post fertilization), (Stocker and Bennett, 2006). Finally, sialylation governs glycoprotein stability and influences cell fate. For example, sialylated β1 integrin suppresses cell adhesion and protects cells against apoptosis (Varki and Schauer, 2009). This protection is essential for cells to undergo an epithelial-to-mesenchymal transition during gastrulation, which is necessary for mesoderm formation and subsequent organogenesis.
Mesoderm formation at gastrulation and the epithelial-to-mesenchymal transition
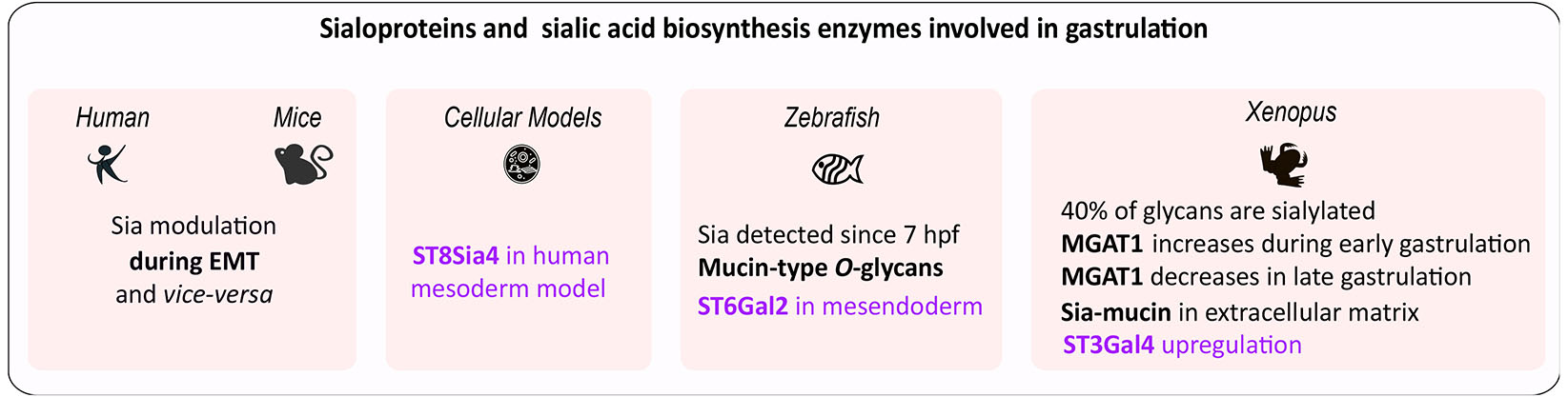
The intermediate cell layer in triploblastic metazoan embryos, mesoderm, is specified during gastrulation, when mesodermal precursors are internalized. This process varies among species. In invertebrates and anamniotes, mesoderm forms through blastoderm folding, as observed in echinoderms, or by rolling, as in amphibians. In amniotes, including birds and mammals, mesodermal cells undergo ingression through the primitive streak (see Nakaya Y., 2008). An epithelial-to-mesenchymal transition (EMT) occurs where cells lose their epithelial traits, modify adhesion by disrupting intercellular contacts and downregulating E-cadherin, and migrate as primary mesenchyme (Hay, 2005). Later, they become secondary epithelial cells and can undergo another EMT to form connective and body tissues. Cycles between EMT and its reverse mesenchymal-to-epithelial transition (MET) also occur during gastrulation and ontogeny (Li et al., 2021; Newton AH, Smith CA, 2024; Thiery et al., 2024). Sialylation significantly influences EMT as proven in tumors (Du et al., 2015). Fig. 3 illustrates the enzymes from the sialic acid biosynthetic pathway involved in gastrulation across various species.
In deyolked zebrafish embryos, the profiles of N-glycan showed stage-dependent variations of complex- and hybrid-type glycans. The presence of sialic acids is detected as early as 7 hpf and increasing at later developmental stages. Complex- and hybrid-type glycans are minor components, and their antenna structures are mainly sialyl LacdiNAc (Siaα2-6GalNAcβ1-4GlcNAc), (Hanzawa et al., 2017). Mucin-type O-glycans are also detected by 7 hpf (Flanagan-Steet and Steet, 2013). ST6GAL2 and ST6GAL2-related genes are expressed at the onset of gastrulation in the mesendoderm (Petit et al., 2010).
In Xenopus, approximately 40% of glycans are sialylated by the end of gastrulation (stage 13), (Qu et al., 2020). The expression of MGAT1 is essential for the synthesis of complex- and hybrid-type N-glycans present in high quantities during early gastrulation (stage 10.5) (Onuma et al., 2013). Gastrulating embryos synthesize Sia-mucin in their extracellular matrix (Johnson, 1977). Regenerating half-embryos showed upregulation of ST3Gal4 at gastrulation, indicating a potential role for this enzyme in pluripotency loss (Sosa et al., 2019).
During differentiation into early progenitor cells of the three embryonic germ layers, human pluripotent stem cells become polysialylated. In differentiated mesoderm and endoderm, NCAM is the primary protein that undergoes polysialylation. Of the two polysialyltransferases responsible for adding polysialic acid chains to target proteins, ST8Sia4 is transcriptionally activated by the Goosecoid network during early mesendoderm differentiation, highlighting this polysialyltransferase gene as a contributor to germ layer formation (Berger et al., 2016).
Organogenesis of mesoderm-derived organs
Mesoderm-derived cells give rise to a large numbers of internal organs in vertebrates, excluding the nervous system (ectoderm) and digestive/respiratory systems (endoderm). Cells of mesodermal origin are abundant throughout vertebrates and represent one of the widest varieties of cell types. They arise from lineages including the paraxial mesoderm (axial skeleton, cartilage, muscle, dermis), intermediate mesoderm (urogenital system), and lateral plate mesoderm (body wall, limbs, cardiovascular system).
In amniotes, extraembryonic mesoderm produces membranes like the yolk sac, amnion, and chorion. (Ferretti and Hadjantonakis, 2019). Noticeably, mesodermal derivatives are also associated with non-mesoderm-derived structures, such as smooth and vascular smooth muscles found in the wall of the gastrointestinal tract (Le Guen et al., 2015), endothelial and mesothelial cells, pericytes, alveolar cells, and lipofibroblasts lining the endothelia of the respiratory tract (Herriges and Morrisey, 2014) that are not discussed in the review.
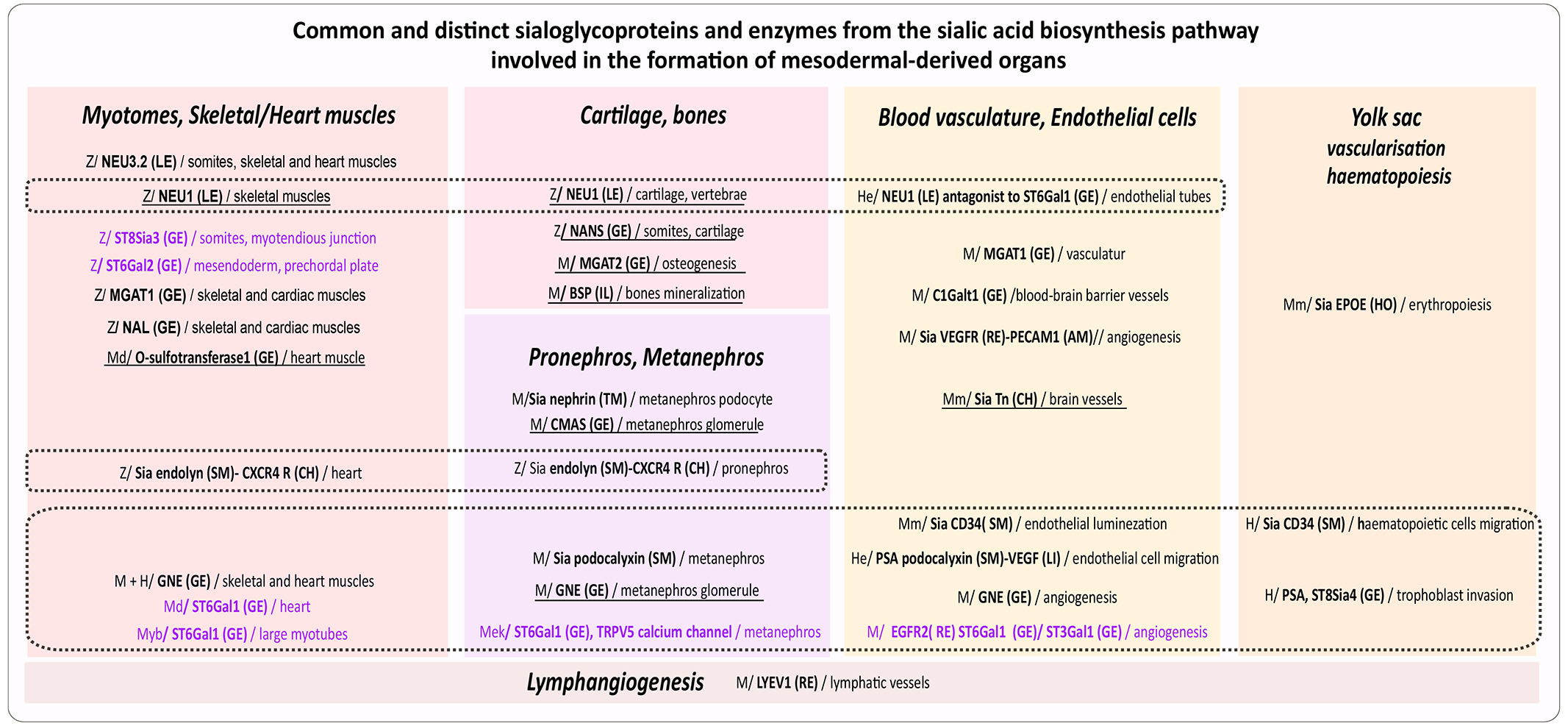
Early morphogenesis frequently begins prior to the completion of gastrulation, and organ development may extend beyond this stage, making it challenging to confine organ morphogenesis strictly to the embryonic period. In aquatic species, organogenesis of mesoderm derivatives typically encompasses both early and later phases, continuing until hatching occurs. In mammals, mesoderm-derived organogenesis is succeeded by sustained fetal growth up to birth. Fig. 4 summarizes sialylated glycoproteins and enzymes from the biosynthesis pathway that are critical for the development of various mesoderm-derived organs across species during the embryonic and early postembryonic periods.
Fig. 4. Common and distinct sialoglycoproteins and enzymes from the sialic acid biosynthesis pathway involved in the formation of mesoderm-derived organs.
Species and models: H: human; He: human endothelial cells; M: mouse; Md: medaka; Mek: mouse embryonic kidney cells; Mm: mammals; Me: mouse embryoids; Myb: mouse myoblasts; Z: zebrafish. Effectors: AM: adhesion molecule; HO: hormone; CH: chemokine; CY: cytokine; GE: Golgi enzyme; IL: integrin domain; LE: lysosome enzyme; RE: receptor; SM: sialomucin; TM: transmembrane protein. Sialyltransferases are purple. Dotted lines demarcate common effectors. Underlined effectors act during late organogenesis (In zebrafish organogenesis starts at 10 hpf and proceeds until hatching at 72 hpf (Hisaoka and Battle, 1958; Kimmel et al., 1995), in medaka it begins at 1 dpf and lasts until hatching at 9 dpf (Iwamatsu, 2004). In mammals, embryonic organogenesis continues with fetal growth until birth. In mice, it begins at 7-8 dpf and continues until 12.5 dpf, followed by fetal growth until 20 dpf (Dyban et al., 1991). In humans, embryonic organogenesis starts at 21 dpf and progresses until approximately 53-58 dpf (8 to 9 weeks post-fertilization) (O’Rahilly and Müller, 2010).
Cartilage and bones
Among the enzymes involved in sialic acid synthesis, NANS is not only essential for brain development but also for skeletal formation. NANS is expressed during early embryonic development in zebrafish, with two duplicated genes for NANS (NANSa and NANSb). Expression begins at 50% epiboly and at 14 to 19 somite stages in the myotome and pharyngeal arch skeleton. Knockdown of NANSa results in an abnormal and complex phenotype regarding skeletal development in the head region at 6 dpf, while no such phenotype is evident for NANSb. The abnormal effects of NANSa knockdown can be partially rescued by the exogenous addition of Neu5Ac. In human, bi-allelic mutations in the NANS gene have been linked to infantile developmental delay and skeletal dysplasia (van Karnebeek et al., 2016).
Two additional enzymes involved in sialylation also influence cartilage formation and bone development. In zebrafish, CRISPR/Cas9 deletion of NEU1 results in abnormal embryogenesis with pleural effusion but not lethality. Neu1-KO fish display reduced body size, muscle atrophy, and vertebral curvature after 8 months, along with decreased expression of bone remodeling genes (runx2a, runx2b, mmp9). These traits closely resemble those seen in Neu1-KO mice and human sialidosis patients (Okada et al., 2020). In mice, knockout of MGAT2 results in increased osteogenic activity just 7 days after birth (Wang et al., 2001).
The Bone SialoProtein (BSP) is a highly phosphorylated and glycosylated secretory protein enriched in Sia, essential for forming the extracellular matrix and ensuring complete mineralization of bones in mice. Removal of terminal sialylation of BSP triggers the downregulation of osteogenesis-related osteocalcin, osteoprotegerin, and vitamin D receptor at the mRNA and protein levels (Malaval et al., 2008; Xu et al., 2017).
Muscles
Sialome imaging in live zebrafish embryos using copper-free click chemistry revealed that sialic acids are present in muscle cells, in addition to ectodermal-derivatives at 24 hpf (Hong et al., 2019). At 30 and 48 hpf, a dense sialylation is observed in intermyotomal boundaries, and at the surface of muscle fiber cells within the myotomes, known to express ST8Sia3 (Agarwal et al., 2015). ST8Sia3 is initially expressed in somites at 20 hpf. Morpholino-knockdown of ST8Sia3 leads to anomalies in somite morphology, including defects in the segment boundary and the myotendinous junction at 35 hpf (Bentrop et al., 2008). The phenotypes might either be somitic or secondary due to axonal defects (Flanagan-Steet and Steet, 2013). The ST6Gal2 gene, which is expressed since the gastrula stage, is found in the mesoderm of the prechordal plate, in early somite, at the borders of the neural plate, with persistent expression in the last formed somite at 24 hpf (Petit et al., 2010). The inactivation of the zebrafish MGAT1 gene, alongside the disruption of complex N-glycans results in skeletal and cardiac muscle defects at 28 and 58 hpf (Hall et al., 2023). Gene targeting experiments using morpholinos against NAL generate skeletal myopathy and cardiac edema at 48hpf, mimicking a human disease phenotype (Wen et al., 2018). Additionally, zebrafish with NEU1 CRISPR/Cas9 deletions exhibit muscle atrophy and reduction in muscle determinants after 8 months (Okada et al., 2020). NEU3.2, analogous to human NEU2, is essential for zebrafish skeletal muscle differentiation; morpholino knockdown leads to major embryonic defects in somites, the heart, and anterior-posterior axis formation (Zizioli et al., 2023). Furthermore, knockdown in zebrafish using morpholino oligonucleotides targeting the sialomucin endolyn (CD164) results in pericardial edema at 48 hpf (Mo et al., 2012).
In medaka embryos, the knockout of O-sulfotransferase Wscd1, responsible for transferring the sulfonyl group to the hydroxy group Sia, caused heart arrhythmias, decreased ventricular contractile force, and reduced levels of cardiac myosin heavy chain at 8 dpf (Ertunc et al., 2022). The medaka ST6Gal1 knockouts demonstrated pronounced cardiac abnormalities between 7 and 16 days post-fertilization (dpf), resulting in mortality at 14–18 dpf. This lethality was reversed by expression of ST6Gal1, ST6Gal2, and ST3Gal4, indicating that sialylation, rather than a specific sialic acid linkage, is critical for proper cardiac development (Omoto et al., 2023).
In zebrafish, the depletion of GNE impairs skeletal muscles, leading to several abnormalities (Livne et al., 2012; Daya et al., 2014). In mice, the disruption of sialic acid synthesis results in embryonic lethality at 8.5 dpf, accompanied by poor differentiation of skeletal and cardiac muscle cells, and nerve cells (Varki, 2017). Homozygous inactivation of the GNE gene in the UDP-GlcNAc 2-epimerase domain causes early embryonic lethality in mice between 8.5 and 9.5 dpf due to a loss of de novo biosynthesis that cannot be compensated by a contribution of maternal sialylated glycoproteins (Schwarzkopf et al., 2002). Heterozygous GNE-deficient mice survived with reduced sialylation. GNE also plays a significant role in skeletal and cardiac muscle development from mouse ESC. Cardiac and muscle cells in GNE -/- develop at the same time as control cells, but cardiac cells degrade rapidly with decreased beating capacity, and skeletal muscle cells are rare (Milman Krentsis et al., 2011). Mutations in the GNE gene are linked to a rare and unique recessive human neuromuscular disorder known as hereditary inclusion body myopathy (HIBM) (Eisenberg et al., 2001; Carrillo et al., 2018), and to sialuria (Leroy et al., 2001).
In mouse myoblasts C2C12 cell line, differentiation depends on a decrease in α2,6-linked Sia. The suppression of ST6Gal1 impacts Notch signaling pathway activity, down-regulates Pax7 expression, triggers earlier myotube fusion, and the suppression of ST6Gal1 (Vergé et al., 2020).
Kidneys
The urinary system in amniotes originates from the intermediate mesoderm. In lower vertebrates, such as fish, the mesonephros is the final kidney. Endolyn (CD164), which associates with the chemokine receptor CXCR4, is expressed in the proximal tubule and distal segments of the kidney. In zebrafish, it is expressed in the developing pronephros. Targeting sialylated endolyn with morpholino oligonucleotides leads to hydrocephaly, body curvature, altered clearance of the pronephric kidney, in addition to pericardial edema at 48 hpf (Mo et al., 2012).
In mammals, the metanephros is the final kidney (with nephrogenesis starting at 11 dpf in mice and 5 weeks in humans (Desgrange and Cereghini, 2015). Studies in rodents reported that the high sialylation of podocalyxin plays a pivotal role in maintaining the specialized epithelial podocyte architecture and facilitating glomerular filtration (Orlando et al., 2001). Deficits in sialylation impair podocyte maturation (Weinhold et al., 2012). Human podocalyxin has a mucin domain with a serine/threonine content for putative O-glycosylation modifications in its extracellular domain and also has five potential N-linked glycosylation sites and three putative glycosaminoglycan sites. Deficits in sialylation impair podocyte maturation and glomerular filtration in the metanephros (Li and Ding, 2019). In homozygous mutated (M712T GNE/MNK mutation) GNE mice, at 17 dpf, the embryos appear normal externally but die within 1 to 3 days after birth, after showing significant muscle deficiencies, severe glomerular hematuria, podocytopathy associated with hyposialylation of podocalyxin, and increased GNE epimerase activity. Interestingly, these phenotypes are rescued through dietary supplementation with ManNAc, a precursor of sialic acid that crosses cellular membranes (Galeano et al., 2007; Quaggin, 2007), suggesting a potential method to mitigate some glomerulopathies associated with hyposialylation (Kakani et al., 2012). Additionally, CMAS knockdown in mice yields mature kidney glomeruli of the inner cortex with enlarged glomerular urinary space one day after birth (Weinhold et al., 2012). These mice exhibit impaired podocyte formation, foot process effacement, and decreased sialylation of nephrin and podocalyxin mirroring features found in patients with nephrin mutations related to Finnish-type congenital nephrotic syndrome.
Klotho, a glycosidase that removes α2,6-linked Sia, modulates the activity of the TRPV5 (transient receptor potential vanilloid type 5) calcium channel to allow transcellular calcium transport in the kidney distal tubule and may be relevant in kidney organogenesis (Cha et al., 2008; Leunissen et al., 2013).
Vasculature
Endothelial cells and angiogenesis
Endothelial precursors develop from splanchnopleural mesoderm into blood vessels, forming a primary capillary plexus that expands into a network (Jain, 2003). The recruitment of pericytes and smooth muscle cells contributes to this network formation (Carmeliet, 2005). This multistep process is regulated by factors such as the vascular endothelial growth factor (VEGF) and its receptors (Coultas et al., 2005). VEGF stimulates angiogenesis (Simons et al., 2016) through its binding to VEGFR2 carrying a2,6-sialylated glycans, via a reinforced electrostatic interaction (Chiodelli et al., 2017; D’Addio et al., 2020; Koch and Claesson-Welsh, 2012). In human umbilical cells (HUVEC), silencing of ST6Gal1 results in ST3Gal1 upregulation and α2,3-sialylation of VEGFR2 that is still able to bind the VEGF ligand. The concomitant silencing of both ST6Gal1 (affecting N-glycans) and ST3Gal1 (affecting O-glycans) abolishes VEGF/VEGFR2 signaling, suggesting that only negatively charged sialic acids, but not their chemical presentation, are required to support VEGFR2 activation (Chiodelli et al., 2017; D’Addio et al., 2020). Losses of 9-O-acetylation of sialic acids are observed, with ST6Gal1 deficiency causing loss on the endothelium (Martin et al., 2002). Overexpression of NEU1 blocks endothelial tube formation, while the overexpression of ST6Gal1 restores it in human pulmonary microvascular and artery endothelial cells (HPMEC, HPAEC) (Lee et al., 2014). α2,6-sialylation also regulates VEGF receptors binding to PECAM1 and promotes endothelial cell migration and angiogenesis sprouting in mice (Imamaki et al., 2018).
A recent study using a GNE loss-of-function mutation related to the de novo biosynthesis of Sia, showed GNE to be important for angiogenesis. A mouse model carrying one of these mutations (P735R in the MNK domain) was also detected in a pediatric macrothrombocytopenia, exhibiting defective angiogenesis and abnormal megakaryocyte accumulation combined with cerebral hemorrhages at 11 dpf and death at 12.5 dpf (Huang et al., 2024).
Inactivation of MGAT1 in mice results in lethality at 9.5 dpf with embryos displaying vasculature defects, in addition to neural tube anomalies and a situs inversus (Ioffe et al., 1997).
The anti-adhesive sialomucin CD34 is involved in lumen formation. It is localized at endothelial contact sites, where it interacts to the underlying actin cytoskeleton to enable cell shape modification and lumen formation. The negatively charged (due to sialic acids) extracellular domains of CD34 create a narrow space, forming a slit through repulsive interactions between opposing surfaces (Robbins and Beitel, 2010; Strilić et al., 2010).
The core 1 β1-3-galactosyltransferase (C1Galt1 or T-synthase) transfers Gal from UDP-Gal to GalNAc-Ser/Thr (Tn antigen) to form the core 1 O-glycan Galβ1-3GalNAc-Ser/Thr (T antigen), which serves as a precursor for extended and branched O-glycans necessary for angiogenesis and prevention of fatal embryonic brain hemorrhage. In wild-type mice, Neu5Acα2-3Galβ1-3GalNAc-Ser/Thr is localized in endothelial and hematopoietic cells during development. Gene-targeted mice lacking T-synthase, which cannot express the sialylated form of Tn antigen, manifest fatal brain hemorrhage at 14 dpf with a chaotic microvascular network in the brain, distorted capillary lumens, and abnormal association of endothelial cells with pericytes (Xia et al., 2004) necessary structures to form the blood-brain barrier (Bauer et al., 2014).
Interestingly, genetic disruption of Notch signaling in mice halts embryonic vascular expansion between 7.5 to 11.5 dpf and is lethal (Hofmann and Iruela-Arispe, 2007). Notch activity is modulated by the β3-N-acetylglucosaminyltransferase radical Fringe which results in a GlcNAcβ1-3Fucose disaccharide that can be further elongated to form a sialylated tetrasaccharide, Siaα2-6Galβ1-4GlcNAcβ1-3Fuc (Moloney et al., 2000a; Moloney et al., 2000b).
Lymphangiogenesis
After their formation, lymphatic endothelium and lymph nodes in mice express LYVE1 at 9.5 days post-fertilization (Alitalo et al., 2005; Oliver and Alitalo, 2005), as do arterial and venous endothelium (Gordon et al., 2008), and undergo a regulation by sialylation on LYVE1 (Nightingale et al., 2009; Schauer, 2009).
Yolk sac endothelium and hematopoiesis in placental species
In placental species, an extraembryonic mesoderm (ExM) develops around the time of gastrulation as a supportive structure (Ross and Boroviak, 2020). In human, it contributes to the formation of the placental chorionic villi which infiltrate the endometrium, connect to the maternal circulation (Pham et al., 2022), and form the extraembryonic vessels in the trophoblast villi.
PSA formed onto N-linked carbohydrate structures by ST8Sia4 are involved in the migration and invasion of human cytotrophoblasts (Hromatka et al., 2013). During early erythropoiesis, PSA are also present on human hematopoietic progenitors and myeloid cells. Studies on st8sia4 knockdown mice demonstrated their importance in the development of hematopoietic progenitors (Drake et al., 2008; Drake et al., 2009).
Sialomucin CD34, expressed on endothelial cells of the yolk sac, enhances trafficking and migration of hematopoietic cells by favoring their binding to E and P selectin on endothelial cells in humans. The lack of CD34 significantly impairs the expansion of hematopoietic progenitor cells in vitro (AbuSamra et al., 2017).
Highly sialylated erythropoietin, produced by the yolk sac around 9 dpf, is crucial for early erythropoiesis following heartbeat activation in mice (Hirano and Suzuki, 2019). Due to its lower receptor affinity and extended serum half-life, highly sialylated erythropoietin more effectively stimulates erythrocyte production (Gross and Lodish, 2006).
Conclusion
Mesodermal cells, which are specified during gastrulation, develop into a broad range of cell types, including those forming the musculoskeletal system, kidneys and the cardiovascular system. The formation of mesoderm is guided by inductive processes that integrate both external factors, such as maternal and environmental influences, and internal self-organizing signals responsible for patterning and morphogenesis. Despite progress, it remains challenging to consistently understand how different mesodermal cell types emerge across various contexts and timeframes. Although the physiological roles of certain sialic acids (in particular, some biosynthetic enzymes) in the embryo have been identified, the exact functions of sialyltransferases remain poorly understood. Ongoing research continues to address the biological impact and mechanistic details of sialic acids, as well as how sialic acid glycoproteins operate. In many cases, gene knockouts affect multiple steps in organ formation, probably because the enzymes involved in sialylation often have overlapping functions with possible compensation. Additionally, clarifying discrepancies between species regarding embryogenesis timing and modalities is necessary. Recent advancements in single-cell 'omics' have facilitated the reconstruction of potential developmental trajectories and dynamics of lineage specification at the cellular level (Ferretti and Hadjantonakis, 2019). The emergence of gastruloids -3D aggregates capable of self-organization and mimicking aspects of gastrulation - holds promise to study these mechanisms beyond the confines of embryos from placental species (Morales et al., 2021; Simpson and Alberio, 2023). Furthermore, organoid systems that replicate organ structures or organs-on-chips derived from stem cells with vascularization capabilities, such as those for the kidney, provide models to enhance our understanding of mammalian organogenesis (Liu et al., 2024; Ryan and Cleaver, 2022). Moreover, a complete model of a human embryo created 14 days after implantation from embryonic stem cells may offer a simpler way to explore early stages of development in accordance with ethical laws (Oldak et al., 2023). All these innovative biological systems could help clarify specific aspects of developmental processes from early and late organogenesis in mesoderm-derived tissues, as well as the precise role of sialylated effectors and enzymes involved in sialylation.
Abbreviations
AcCoA, acetyl-coenzyme A ; CMAH, N-acetylneuraminic acid hydroxylase ; CMAS or CSS, CMP-sialic acid synthetase ; CMP, cytidine monophosphate ; dpf, days post-fertilization ; GalNAc, N-acetylgalactosamine ; GlcNAc, N-acetylglucosamine ; GNE/MNK, glucosamine UDP-GlcNAc-2-epimerase/N-acetylmannosamine kinase ; hpf, hours post-fertilization ; Kdn, 2-keto-3-deoxy-D-glycero-D-galacto-nonulosonic acid ; ManNAc, N-acetyl-mannosamine ; NANP, N-acetylneuraminate-9-phosphatase ; NAL, N-acetylneuraminate lyase ; NANS, Neu5Ac-9-P synthase ; Neu, neuraminic acid ; NEU, neuraminidase ; Neu5Ac, N-acetylneuraminic acid ; Neu5Gc, N-glycolylneuraminic acid ; NCAM, neural cell adhesion molecule ; PSA, polysialic acids ; Sia, sialic acids ; sLex, sialyl-Lewis(x) ; sLea, sialyl-Lewis(a) ; ST8Sia2, Alpha-N-Acetyl-Neuraminide Alpha-2,8-Sialyltransferase 2 ; ST8Sia4, Alpha-N-Acetyl-Neuraminide Alpha-2,8-Sialyltransferase 4 ; UDP, uridine diphosphate ;Declarations
Conflicts of interest
The authors declare no conflict of interest.
Author contributions
Conceptualization: I.F., A.H.L and K.C. Writing: I.F., P.D., A.F., M.M., A.M., Y.U., N.Y., C.M., J.F.B., A.H.L. and K.C.
Funding
This work was supported by the CNRS and the University of Lille.
References
Abeln M., Albers I., Peters-Bernard U., Flächsig-Schulz K., Kats E., Kispert A., Tomlinson S., Gerardy-Schahn R., Münster-Kühnel A., Weinhold B. (2019). Sialic acid is a critical fetal defense against maternal complement attack. Journal of Clinical Investigation 129: 422-436.
AbuSamra D. B., Aleisa F. A., Al-Amoodi A. S., Jalal Ahmed H. M., Chin C. J., Abuelela A. F., Bergam P., Sougrat R., Merzaban J. S. (2017). Not just a marker: CD34 on human hematopoietic stem/progenitor cells dominates vascular selectin binding along with CD44. Blood Advances 1: 2799-2816.
Agarwal P., Beahm B. J., Shieh P., Bertozzi C. R. (2015). Systemic Fluorescence Imaging of Zebrafish Glycans with Bioorthogonal Chemistry. Angewandte Chemie International Edition 54: 11504-11510.
Alitalo K., Tammela T., Petrova T. V. (2005). Lymphangiogenesis in development and human disease. Nature 438: 946-953.
Altman M. O., Gagneux P. (2019). Absence of Neu5Gc and Presence of Anti-Neu5Gc Antibodies in Humans—An Evolutionary Perspective. Frontiers in Immunology 10: 789.
Aoki K., Perlman M., Lim J.M., Cantu R., Wells L., Tiemeyer M. (2007). Dynamic Developmental Elaboration of N-Linked Glycan Complexity in the Drosophila melanogaster Embryo. Journal of Biological Chemistry 282: 9127-9142.
Bacic A., Kahane I., Zuckerman B. M. (1990). Panagrellus redivivus and Caenorhabditis elegans: Evidence for the absence of sialic acids. Experimental Parasitology 71: 483-488.
Bauer H.C., Krizbai I. A., Bauer H., Traweger A. (2014). "You Shall Not Pass"-tight junctions of the blood brain barrier. Frontiers in Neuroscience 8: 1-21.
Bentrop J., Marx M., Schattschneider S., Rivera‐Milla E., Bastmeyer M. (2008). Molecular evolution and expression of zebrafish St8SiaIII, an alpha‐2,8‐sialyltransferase involved in myotome development. Developmental Dynamics 237: 808-818.
Berger R. P., Sun Y. H., Kulik M., Lee J. K., Nairn A. V., Moremen K. W., Pierce M., Dalton S. (2016). ST8SIA4-Dependent Polysialylation is Part of a Developmental Program Required for Germ Layer Formation from Human Pluripotent Stem Cells. Stem Cells 34: 1742-1752.
Bork K., Weidemann W., Berneck B., Kuchta M., Bennmann D., Thate A., Huber O., Gnanapragassam V. S., Horstkorte R. (2017). The expression of sialyltransferases is regulated by the bioavailability and biosynthesis of sialic acids. Gene Expression Patterns 23-24: 52-58.
Born G. V. R., Palinski W., (1985). Unusually high concentrations of sialic acids on the surface of vascular endothelia. British Journal of Experimental Pathology 66: 543-549.
Carrillo N., Malicdan M. C., Huizing M. (2018). GNE Myopathy: Etiology, Diagnosis, and Therapeutic Challenges. Neurotherapeutics 15: 900-914.
Carmeliet P. (2005). Angiogenesis in life, disease and medicine. Nature 438: 932-936.
Carvalho A.S., Harduin-Lepers A., Magalhães A., Machado E., Mendes N., Costa L.T., Matthiesen R., Almeida R., Costa J., Reis C.A. (2010). Differential expression of α-2,3-sialyltransferases and α-1,3/4-fucosyltransferases regulates the levels of sialyl Lewis a and sialyl Lewis x in gastrointestinal carcinoma cells. The International Journal of Biochemistry & Cell Biology 42: 80-89.
Cha S.K., Ortega B., Kurosu H., Rosenblatt K. P., Kuro-o M., Huang C.L. (2008). Removal of sialic acid involving Klotho causes cell-surface retention of TRPV5 channel via binding to galectin-1. Proceedings of the National Academy of Sciences 105: 9805-9810.
Chang L.Y., Mir A.M., Thisse C., Guérardel Y., Delannoy P., Thisse B., Harduin-Lepers A. (2009). Molecular cloning and characterization of the expression pattern of the zebrafish α2, 8-sialyltransferases (ST8Sia) in the developing nervous system. Glycoconjugate Journal 26: 263-275.
Chen X., Varki A. (2010). Advances in the Biology and Chemistry of Sialic Acids. ACS Chemical Biology 5: 163-176.
Chiodelli P., Rezzola S., Urbinati C., Federici Signori F., Monti E., Ronca R., Presta M., Rusnati M. (2017). Contribution of vascular endothelial growth factor receptor-2 sialylation to the process of angiogenesis. Oncogene 36: 6531-6541.
Cohen M., Varki A. (2010). The Sialome—Far More Than the Sum of Its Parts. OMICS: A Journal of Integrative Biology 14: 455-464.
Coultas L., Chawengsaksophak K., Rossant J. (2005). Endothelial cells and VEGF in vascular development. Nature 438: 937-945.
D’Addio M., Frey J., Otto V. I. (2020). The manifold roles of sialic acid for the biological functions of endothelial glycoproteins. Glycobiology 30: 490-499.
Daya A., Vatine G. D., Becker-Cohen M., Tal-Goldberg T., Friedmann A., Gothilf Y., Du S. J., Mitrani-Rosenbaum S. (2014). Gne depletion during zebrafish development impairs skeletal muscle structure and function. Human Molecular Genetics 23: 3349-3361.
Desgrange A., Cereghini S. (2015). Nephron Patterning: Lessons from Xenopus, Zebrafish, and Mouse Studies. Cells 4: 483-499.
Drake P. M., Nathan J. K., Stock C. M., Chang P. V., Muench M. O., Nakata D., Reader J. R., Gip P., Golden K. P. K., Weinhold B., Gerardy-Schahn R., Troy F. A., Bertozzi C. R. (2008). Polysialic Acid, a Glycan with Highly Restricted Expression, Is Found on Human and Murine Leukocytes and Modulates Immune Responses. The Journal of Immunology 181: 6850-6858.
Drake P. M., Stock C. M., Nathan J. K., Gip P., Golden K. P. K., Weinhold B., Gerardy-Schahn R., Bertozzi C. R. (2009). Polysialic acid governs T-cell development by regulating progenitor access to the thymus. Proceedings of the National Academy of Sciences 106: 11995-12000.
Du J., Hong S., Dong L., Cheng B., Lin L., Zhao B., Chen Y.G., Chen X. (2015). Dynamic Sialylation in Transforming Growth Factor-β (TGF-β)-induced Epithelial to Mesenchymal Transition. Journal of Biological Chemistry 290: 12000-12013.
Dyban A. P., Puchkov V. F., Samoshkina N. A., Khozhai L. I., Chebotar' N. A., Baranov V. S., (1991). Laboratory Mammals: Mouse (Mus musculus), Rat (Rattus norvegicus), Rabbit (Oryctolagus cuniculus), and Golden Hamster (Cricetus auratus). In Animal Species for Developmental Studies. (Ed. Dettlaff T. A., Vassetzky S. G., ) Springer, Boston, MA.
Eisenberg I., Avidan N., Potikha T., Hochner H., Chen M., Olender T., Barash M., Shemesh M., Sadeh M., Grabov-Nardini G., Shmilevich I., Friedmann A., Karpati G., Bradley W. G., Baumbach L., Lancet D., Asher E. B., Beckmann J. S., Argov Z., Mitrani-Rosenbaum S. (2001). The UDP-N-acetylglucosamine 2-epimerase/N-acetylmannosamine kinase gene is mutated in recessive hereditary inclusion body myopathy. Nature Genetics 29: 83-87.
Ertunc N., Phitak T., Wu D., Fujita H., Hane M., Sato C., Kitajima K. (2022). Sulfation of sialic acid is ubiquitous and essential for vertebrate development. Scientific Reports 12: 12496.
Ferretti E., Hadjantonakis A.K. (2019). Mesoderm specification and diversification: from single cells to emergent tissues. Current Opinion in Cell Biology 61: 110-116.
Flanagan-Steet H. R., Steet R. (2013). “Casting” light on the role of glycosylation during embryonic development: Insights from zebrafish. Glycoconjugate Journal 30: 33-40.
Frappaolo A., Sechi S., Kumagai T., Robinson S., Fraschini R., Karimpour-Ghahnavieh A., Belloni G., Piergentili R., Tiemeyer K. H., Tiemeyer M., Giansanti M. G. (2017). COG7 deficiency in Drosophila generates multifaceted developmental, behavioral and protein glycosylation phenotypes. Journal of Cell Science 130: 3637-3649.
Galeano B., Klootwijk R., Manoli I., Sun M.S., Ciccone C., Darvish D., Starost M. F., Zerfas P. M., Hoffmann V. J., Hoogstraten-Miller S., Krasnewich D. M., Gahl W. A., Huizing M. (2007). Mutation in the key enzyme of sialic acid biosynthesis causes severe glomerular proteinuria and is rescued by N-acetylmannosamine. Journal of Clinical Investigation 117: 1585-1594.
Galuska C., Lütteke T., Galuska S. (2017). Is Polysialylated NCAM Not Only a Regulator during Brain Development But also during the Formation of Other Organs?. Biology 6: 27.
Ghosh S., (2020). Sialic acid and biology of life: An introduction. In Sialic Acids and Sialoglycoconjugates in the Biology of Life, Health and Disease. Elsevier Inc..
Gordon E. J., Gale N. W., Harvey N. L., (2008). Expression of the hyaluronan receptor LYVE-1 is not restricted to the lymphatic vasculature; LYVE-1 is also expressed on embryonic blood vessels. Developmental Dynamics 237: 1901-1909.
Gross A. W., Lodish H. F. (2006). Cellular Trafficking and Degradation of Erythropoietin and Novel Erythropoiesis Stimulating Protein (NESP). Journal of Biological Chemistry 281: 2024-2032.
Le Guen L., Marchal S., Faure S., de Santa Barbara P. (2015). Mesenchymal–epithelial interactions during digestive tract development and epithelial stem cell regeneration. Cellular and Molecular Life Sciences 72: 3883-3896.
Hall M. K., Hatchett C. J., Shalygin S., Azadi P., Schwalbe R. A. (2023). Reduction in N-Acetylglucosaminyltransferase-I Activity Decreases Survivability and Delays Development of Zebrafish. Current Issues in Molecular Biology 45: 9165-9180.
Hanzawa K., Suzuki N., Natsuka S. (2017). Structures and developmental alterations of N-glycans of zebrafish embryos. Glycobiology 27: 228-245.
Harduin-Lepers A., Mollicone R., Delannoy P., Oriol R. (2005). The animal sialyltransferases and sialyltransferase-related genes: a phylogenetic approach. Glycobiology 15: 805-817.
Harduin-Lepers A. (2023). The vertebrate sialylation machinery: structure-function and molecular evolution of GT-29 sialyltransferases. Glycoconjugate Journal 40: 473-492.
Hay E. D. (2005). The mesenchymal cell, its role in the embryo, and the remarkable signaling mechanisms that create it. Developmental Dynamics 233: 706-720.
Henrissat B., Bairoch A. (1996). Updating the sequence-based classification of glycosyl hydrolases. Biochemical Journal 316: 695-696.
Herriges M., Morrisey E. E. (2014). Lung development: orchestrating the generation and regeneration of a complex organ. Development 141: 502-513.
Hirano I., Suzuki N. (2019). The Neural Crest as the First Production Site of the Erythroid Growth Factor Erythropoietin. Frontiers in Cell and Developmental Biology 7: 105.
Hisaoka K. K., Battle H. I. (1958). The normal developmental stages of the zebrafish, brachydanio rerio (hamilton‐buchanan). Journal of Morphology 102: 311-327.
Hofmann J. J., Iruela-Arispe M. L. (2007). Notch Signaling in Blood Vessels. Circulation Research 100: 1556-1568.
Hong S., Sahai‐Hernandez P., Chapla D. G., Moremen K. W., Traver D., Wu P. (2019). Direct Visualization of Live Zebrafish Glycans via Single‐Step Metabolic Labeling with Fluorophore‐Tagged Nucleotide Sugars. Angewandte Chemie International Edition 58: 14327-14333.
Hromatka B. S., Drake P. M., Kapidzic M., Stolp H., Goldfien G. A., Shih I.M., Fisher S. J. (2013). Polysialic acid enhances the migration and invasion of human cytotrophoblasts. Glycobiology 23: 593-602.
Huang L., Kondo Y., Cao L., Han J., Li T., Zuo B., Yang F., Li Y., Ma Z., Bai X., Jiang M., Ruan C., Xia L. (2024). Novel GNE missense variants impair de novo sialylation and cause defective angiogenesis in the developing brain in mice. Blood Advances 8: 991-1001.
Imamaki R., Ogawa K., Kizuka Y., Komi Y., Kojima S., Kotani N., Honke K., Honda T., Taniguchi N., Kitazume S. (2018). Glycosylation controls cooperative PECAM-VEGFR2-β3 integrin functions at the endothelial surface for tumor angiogenesis. Oncogene 37: 4287-4299.
Ioffe E., Liu Y., Stanley P. (1997). Complex N -glycans in Mgat1 null preimplantation embryos arise from maternal Mgat1 RNA. Glycobiology 7: 913-919.
Iwamatsu T. (2004). Stages of normal development in the medaka Oryzias latipes. Mechanisms of Development 121: 605-618.
Jain R. K. (2003). Molecular regulation of vessel maturation. Nature Medicine 9: 685-693.
Johnson K. E. (1977). Extracellular matrix synthesis in blastula and gastrula stages of normal and hybrid frog embryos: iv. biochemical and autoradiographic observations on fucose-, glucose-, and mannose-labelled materials. Journal of Cell Science 32: 109-136.
Kajiura H., Hamaguchi Y., Mizushima H., Misaki R., Fujiyama K. (2015). Sialylation potentials of the silkworm, Bombyx mori ; B. mori possesses an active α2,6-sialyltransferase. Glycobiology 25: 1441-1453.
Kakani S., Yardeni T., Poling J., Ciccone C., Niethamer T., Klootwijk E. D., Manoli I., Darvish D., Hoogstraten-Miller S., Zerfas P., Tian E., Ten Hagen K. G., Kopp J. B., Gahl W. A., Huizing M. (2012). The Gne M712T Mouse as a Model for Human Glomerulopathy. The American Journal of Pathology 180: 1431-1440.
Kimmel C. B., Ballard W. W., Kimmel S. R., Ullmann B., Schilling T. F. (1995). Stages of embryonic development of the zebrafish. Developmental Dynamics 203: 253-310.
Kitazume S., Imamaki R., Ogawa K., Komi Y., Futakawa S., Kojima S., Hashimoto Y., Marth J. D., Paulson J. C., Taniguchi N. (2010). α2,6-Sialic Acid on Platelet Endothelial Cell Adhesion Molecule (PECAM) Regulates Its Homophilic Interactions and Downstream Antiapoptotic Signaling. Journal of Biological Chemistry 285: 6515-6521.
Koch S., Claesson-Welsh L., (2012). Signal Transduction by VEGFRs. Cold Spring Harbor Perspectives in Medicine 1-21.
Koles K., Irvine K. D., Panin V. M. (2004). Functional Characterization of Drosophila Sialyltransferase. Journal of Biological Chemistry 279: 4346-4357.
Lee C., Liu A., Miranda-Ribera A., Hyun S. W., Lillehoj E. P., Cross A. S., Passaniti A., Grimm P.R., Kim B.Y., Welling P. A., Madri J. A., DeLisser H. M., Goldblum S. E. (2014). NEU1 Sialidase Regulates the Sialylation State of CD31 and Disrupts CD31-driven Capillary-like Tube Formation in Human Lung Microvascular Endothelia. Journal of Biological Chemistry 289: 9121-9135.
Lepers A., Cacan R., Verbert A. (1990). Permeabilized cells as a way of gaining access to intracellular organelles: an approach to glycosylation reactions. Biochimie 72: 1-5.
Lepers A., Shaw L., Cacan R., Schauer R., Montreuil J., Verbert A. (1989). Transport of CMP‐ N ‐glycoloylneuraminic acid into mouse liver Golgi vesicles. FEBS Letters 250: 245-250.
Leroy J. G., Seppala R., Huizing M., Dacremont G., De Simpel H., Van Coster R. N., Orvisky E., Krasnewich D. M., Gahl W. A. (2001). Dominant Inheritance of Sialuria, an Inborn Error of Feedback Inhibition. The American Journal of Human Genetics 68: 1419-1427.
Leunissen E. H.P., Nair A. V., Büll C., Lefeber D. J., van Delft F. L., Bindels R. J.M., Hoenderop J. G.J. (2013). The Epithelial Calcium Channel TRPV5 Is Regulated Differentially by Klotho and Sialidase. Journal of Biological Chemistry 288: 29238-29246.
Li C.H., Hsu T.I., Chang Y.C., Chan M.H., Lu P.J., Hsiao M. (2021). Stationed or Relocating: The Seesawing EMT/MET Determinants from Embryonic Development to Cancer Metastasis. Biomedicines 9: 1265.
Li F., Ding J. (2019). Sialylation is involved in cell fate decision during development, reprogramming and cancer progression. Protein & Cell 10: 550-565.
Li J., Dai G., Cheng Y.B., Qi X., Geng M.Y. (2011). Polysialylation promotes neural cell adhesion molecule-mediated cell migration in a fibroblast growth factor receptor-dependent manner, but independent of adhesion capability. Glycobiology 21: 1010-1018.
Liu Q., Yue L., Deng J., Tan Y., Wu C. (2024). Progress and breakthroughs in human kidney organoid research. Biochemistry and Biophysics Reports 39: 101736.
Livne H., Avital T., Ruppo S., Harazi A., Mitrani-Rosenbaum S., Daya A. (2022). Generation and characterization of a novel gne Knockout Model in Zebrafish. Frontiers in Cell and Developmental Biology 10: 976111.
Lombard V., Golaconda Ramulu H., Drula E., Coutinho P. M., Henrissat B. (2014). The carbohydrate-active enzymes database (CAZy) in 2013. Nucleic Acids Research 42: D490-D495.
Ma Y., Kirby B. J., Fairbridge N. A., Karaplis A. C., Lanske B., Kovacs C. S. (2017). FGF23 is not required to regulate fetal phosphorus metabolism but exerts effects within 12 hours after birth. Endocrinology 158: 252-263.
Malaval L., Wade-Guéye N. M., Boudiffa M., Fei J., Zirngibl R., Chen F., Laroche N., Roux J.P., Burt-Pichat B., Duboeuf F., Boivin G., Jurdic P., Lafage-Proust M.H., Amédée J., Vico L., Rossant J., Aubin J. E. (2008). Bone sialoprotein plays a functional role in bone formation and osteoclastogenesis. The Journal of Experimental Medicine 205: 1145-1153.
Martin L. T., Marth J. D., Varki A., Varki N. M. (2002). Genetically Altered Mice with Different Sialyltransferase Deficiencies Show Tissue-specific Alterations in Sialylation and Sialic Acid 9-O-Acetylation. Journal of Biological Chemistry 277: 32930-32938.
Milman Krentsis I., Sela I., Eiges R., Blanchard V., Berger M., Becker Cohen M., Mitrani-Rosenbaum S. (2011). GNE Is Involved in the Early Development of Skeletal and Cardiac Muscle. PLoS ONE 6: e21389.
Miyata S., Sato C., Kumita H., Toriyama M., Vacquier V. D., Kitajima K. (2006). Flagellasialin: a novel sulfated 2,9-linked polysialic acid glycoprotein of sea urchin sperm flagella. Glycobiology 16: 1229-1241.
Mo D., Ihrke G., Costa S. A., Brilli L., Labilloy A., Halfter W., Cosentino C. C., Hukriede N. A., Weisz O. A. (2012). Apical Targeting and Endocytosis of the Sialomucin Endolyn are Essential for Establishment of Zebrafish Pronephric Kidney Function. Journal of Cell Science 125: 5546-5554.
Moloney D. J., Panin V. M., Johnston S. H., Chen J., Shao L., Wilson R., Wang Y., Stanley P., Irvine K. D., Haltiwanger R. S., Vogt T. F. (2000a). Fringe is a glycosyltransferase that modifies Notch. Nature 406: 369-375.
Moloney D. J., Shair L. H., Lu F. M., Xia J., Locke R., Matta K. L., Haltiwanger R. S. (2000b). Mammalian Notch1 Is Modified with Two Unusual Forms ofO-Linked Glycosylation Found on Epidermal Growth Factor-like Modules. Journal of Biological Chemistry 275: 9604-9611.
Mondal N., Dykstra B., Lee J., Ashline D. J., Reinhold V. N., Rossi D. J., Sackstein R. (2018). Distinct human α(1,3)-fucosyltransferases drive Lewis-X/sialyl Lewis-X assembly in human cells. Journal of Biological Chemistry 293: 7300-7314.
Monti E., Bonten; E., D'Azzo A., Bresciani R., Venerando B., Borsani G., Schauer R., Tettamanti G. (2010). Sialidases in vertebrates: a family of enzymes tailored for several cell functions.. In Advances in Carbohydrate Chemistry and Biochemistry. (Ed. Horton D.) Elsevier.
Morales J. S., Raspopovic J., Marcon L. (2021). From embryos to embryoids: How external signals and self-organization drive embryonic development. Stem Cell Reports 16: 1039-1050.
Nakaya Y., Sheng G. (2008). Epithelial to mesenchymal transition during gastrulation: An embryological view. Development, Growth & Differentiation 50: 755-766.
Newton A. H., Smith C. A. (2024). Resolving the mechanisms underlying epithelial‐to‐mesenchymal transition of the lateral plate mesoderm. Genesis 62: e23531.
Nightingale T. D., Frayne M. E.F., Clasper S., Banerji S., Jackson D. G. (2009). A Mechanism of Sialylation Functionally Silences the Hyaluronan Receptor LYVE-1 in Lymphatic Endothelium. Journal of Biological Chemistry 284: 3935-3945.
O'Rahilly R., Müller F. (2010). Developmental Stages in Human Embryos: Revised and New Measurements. Cells Tissues Organs 192: 73-84.
Okada K., Takase R., Hamaoka Y., Honda A., Ikeda A., Hokazono Y., Maeda Y., Hayasaka O., Kotani T., Komatsu M., Shiozaki K. (2020). Establishment and characterization of Neu1-knockout zebrafish and its abnormal clinical phenotypes. Biochemical Journal 477: 2841-2857.
Oldak B., Wildschutz E., Bondarenko V., Comar M.Y., Zhao C., Aguilera-Castrejon A., Tarazi S., Viukov S., Pham T. X. A., Ashouokhi S., Lokshtanov D., Roncato F., Ariel E., Rose M., Livnat N., Shani T., Joubran C., Cohen R., Addadi Y., Chemla M., Kedmi M., Keren-Shaul H., Pasque V., Petropoulos S., Lanner F., Novershtern N., Hanna J. H. (2023). Complete human day 14 post-implantation embryo models from naive ES cells. Nature 622: 562-573.
Oliver G., Alitalo K. (2005). THE LYMPHATIC VASCULATURE: Recent Progress and Paradigms. Annual Review of Cell and Developmental Biology 21: 457-483.
Omoto T., Wu D., Maruyama E., Tajima K., Hane M., Sato C., Kitajima K. (2023). Forced expression of α2,3-sialyltransferase IV rescues impaired heart development in α2,6-sialyltransferase I-deficient medaka. Biochemical and Biophysical Research Communications 649: 62-70.
Ono S., Hane M., Kitajima K., Sato C. (2012). Novel Regulation of Fibroblast Growth Factor 2 (FGF2)-mediated Cell Growth by Polysialic Acid. Journal of Biological Chemistry 287: 3710-3722.
Onuma Y., Tateno H., Tsuji S., Hirabayashi J., Ito Y., Asashima M. (2013). A Lectin-Based Glycomic Approach to Identify Characteristic Features of Xenopus Embryogenesis. PLoS ONE 8: e56581.
Orlando R. A., Takeda T., Zak B., Schmieder S., Benoit V. M., McQuistan T., Furthmayr H., Farquhar M. G. (2001). The Glomerular Epithelial Cell Anti-Adhesin Podocalyxin Associates with the Actin Cytoskeleton through Interactions with Ezrin. Journal of the American Society of Nephrology 12: 1589-1598.
Paschinger K., Wilson I. B. H. (2019). Comparisons of N-glycans across invertebrate phyla. Parasitology 146: 1733-1742.
Petit D., Mir A.M., Petit J.M., Thisse C., Delannoy P., Oriol R., Thisse B., Harduin-Lepers A. (2010). Molecular Phylogeny and Functional Genomics of β-Galactoside α2,6-Sialyltransferases That Explain Ubiquitous Expression of st6gal1 Gene in Amniotes. Journal of Biological Chemistry 285: 38399-38414.
Pham T. X. A., Panda A., Kagawa H., To S. K., Ertekin C., Georgolopoulos G., van Knippenberg S. S.F.A., Allsop R. N., Bruneau A., Chui J. S.H., Vanheer L., Janiszewski A., Chappell J., Oberhuemer M., Tchinda R. S., Talon I., Khodeer S., Rossant J., Lluis F., David L., Rivron N., Balaton B. P., Pasque V. (2022). Modeling human extraembryonic mesoderm cells using naive pluripotent stem cells. Cell Stem Cell 29: 1346-1365.e10.
Qu Y., Dubiak K. M., Peuchen E. H., Champion M. M., Zhang Z., Hebert A. S., Wright S., Coon J. J., Huber P. W., Dovichi N. J. (2020). Quantitative capillary zone electrophoresis-mass spectrometry reveals the N -glycome developmental plan during vertebrate embryogenesis. Molecular Omics 16: 210-220.
Quaggin S. E. (2007). Sizing up sialic acid in glomerular disease. Journal of Clinical Investigation 117: 1480-1483.
Robbins R. M., Beitel G. J. (2010). Vascular Lumen Formation: Negativity Will Tear Us Apart. Current Biology 20: R973-R975.
Ross C., Boroviak T. E. (2020). Origin and function of the yolk sac in primate embryogenesis. Nature Communications 11: 3760.
Ryan A. R., Cleaver O. (2022). Plumbing our organs: Lessons from vascular development to instruct lab generated tissues. In Mouse Models of Development and Disease. Elsevier.
Sato C., Kitajima K. (2013). Disialic, oligosialic and polysialic acids: distribution, functions and related disease. Journal of Biochemistry 154: 115-136.
Sato C., Kitajima K. (2021). Polysialylation and disease. Molecular Aspects of Medicine 79: 100892.
Schachter H. (2000). The joys of HexNAc. The synthesis and function of N-andO-glycan branches. Glycoconjugate Journal 17: 465-483.
Schauer R. (1982). Chemistry, Metabolism, and Biological Functions of Sialic Acids. In Advances in Carbohydrate Chemistry and Biochemistry. Elsevier.
Schauer R., Sommer U., Krüger D., van Unen H., Traving C. (1999). The Terminal Enzymes of Sialic Acid Metabolism: Acylneuraminate Pyruvate-Lyases. Bioscience Reports 19: 373-383.
Schauer R. (2009). Sialic acids as regulators of molecular and cellular interactions. Current Opinion in Structural Biology 19: 507-514.
Schauer R., Kamerling J. P., (2018). Exploration of the Sialic Acid World. Elsevier Inc..
Schnaar R. L., Gerardy-Schahn R., Hildebrandt H. (2014). Sialic Acids in the Brain: Gangliosides and Polysialic Acid in Nervous System Development, Stability, Disease, and Regeneration. Physiological Reviews 94: 461-518.
Schwarzkopf M., Knobeloch K.P., Rohde E., Hinderlich S., Wiechens N., Lucka L., Horak I., Reutter W., Horstkorte R. (2002). Sialylation is essential for early development in mice. Proceedings of the National Academy of Sciences 99: 5267-5270.
Simons M., Gordon E., Claesson-Welsh L. (2016). Mechanisms and regulation of endothelial VEGF receptor signalling. Nature Reviews Molecular Cell Biology 17: 611-625.
Simpson L., Alberio R. (2023). Interspecies control of development during mammalian gastrulation. Emerging Topics in Life Sciences 7: 397-408.
Sosa E. A., Moriyama Y., Ding Y., Tejeda-Muñoz N., Colozza G., De Robertis E. M. (2019). Transcriptome analysis of regeneration during Xenopus laevis experimental twinning. The International Journal of Developmental Biology 63: 301-309.
Stocker P. J., Bennett E. S. (2006). Differential Sialylation Modulates Voltage-gated Na+ Channel Gating throughout the Developing Myocardium. The Journal of General Physiology 127: 253-265.
Strilić B., Eglinger J., Krieg M., Zeeb M., Axnick J., Babál P., Müller D. J., Lammert E. (2010). Electrostatic Cell-Surface Repulsion Initiates Lumen Formation in Developing Blood Vessels. Current Biology 20: 2003-2009.
Teppa R., Petit D., Plechakova O., Cogez V., Harduin-Lepers A. (2016). Phylogenetic-Derived Insights into the Evolution of Sialylation in Eukaryotes: Comprehensive Analysis of Vertebrate β-Galactoside α2,3/6-Sialyltransferases (ST3Gal and ST6Gal). International Journal of Molecular Sciences 17: 1-20.
Thiery J. P., Sheng G., Shu X., Runyan R. (2024). How studies in developmental epithelial-mesenchymal transition and mesenchymal-epithelial transition inspired new research paradigms in biomedicine. Development 151: dev200128.
Troy F. A. (1992). Polysialylation: from bacteria to brains. Glycobiology 2: 5-23.
van Karnebeek C. D. M., Bonafé L., Wen X.Y., Tarailo-Graovac M., Balzano S., Royer-Bertrand B., Ashikov A., Garavelli L., Mammi I., Turolla L., Breen C., Donnai D., Cormier-Daire V., Heron D., Nishimura G., Uchikawa S., Campos-Xavier B., Rossi A., Hennet T., Brand-Arzamendi K., Rozmus J., Harshman K., Stevenson B. J., Girardi E., Superti-Furga G., Dewan T., Collingridge A., Halparin J., Ross C. J., Van Allen M. I., Rossi A., Engelke U. F., Kluijtmans L. A. J., van der Heeft E., Renkema H., de Brouwer A., Huijben K., Zijlstra F., Heise T., Boltje T., Wasserman W. W., Rivolta C., Unger S., Lefeber D. J., Wevers R. A., Superti-Furga A. (2016). NANS-mediated synthesis of sialic acid is required for brain and skeletal development. Nature Genetics 48: 777-784.
Varki A. (2017a). Biological roles of glycans. Glycobiology 27: 3-49.
Varki A., Gagneux P. (2012). Multifarious roles of sialic acids in immunity. Annals of the New York Academy of Sciences 1253: 16-36.
Varki A., Hooshmand F., Diaz S., Varki N. M., Hedrick S. M. (1991). Developmental abnormalities in transgenic mice expressing a sialic acid-specific 9-O-acetylesterase. Cell 65: 65-74.
Varki A., Schauer R., (2009). Sialic Acids. In Essentials of Glycobiology. (Ed. Varki A., Cummings R. D., Esko J. D., ) Cold Spring Harbor Laboratory Press, Cold Spring Harbor (NY).
(2015). Essentials of Glycobiology. (Ed. Varki A., Cummings R. D., Esko J. D., Stanley P., Hart G. W., Aebi M., Darvill A. G., Kinoshita T., Packer N. H., ) Cold Spring Harbor Laboratory Press, NY.
Vergé C., Bouchatal A., Chirat F., Guérardel Y., Maftah A., Petit J.M. (2020). Involvement of ST6Gal I‐mediated α2,6 sialylation in myoblast proliferation and differentiation. FEBS Open Bio 10: 56-69.
Wang Y., Tan J., Sutton-Smith M., Ditto D., Panico M., Campbell R. M., Varki N. M., Long J. M., Jaeken J., Levinson S. R., Wynshaw-Boris A., Morris H. R., Le D., Dell A., Schachter H., Marth J. D. (2001). Modeling human congenital disorder of glycosylation type IIa in the mouse: conservation of asparagine-linked glycan-dependent functions in mammalian physiology and insights into disease pathogenesis. Glycobiology 11: 1051-1070.
Weinhold B., Sellmeier M., Schaper W., Blume L., Philippens B., Kats E., Bernard U., Galuska S. P., Geyer H., Geyer R., Worthmann K., Schiffer M., Groos S., Gerardy-Schahn R., Münster-Kühnel A. K. (2012). Deficits in Sialylation Impair Podocyte Maturation. Journal of the American Society of Nephrology 23: 1319-1328.
Wen X.Y., Tarailo-Graovac M., Brand-Arzamendi K., Willems A., Rakic B., Huijben K., Da Silva A., Pan X., El-Rass S., Ng R., Selby K., Philip A. M., Yun J., Ye X. C., Ross C. J., Lehman A. M., Zijlstra F., Bakar A. A., Drögemöller B., Moreland J., Wasserman W. W., Vallance H., van Scherpenzeel M., Karbassi F., Hoskings M., Engelke U., de Brouwer A., Wevers R. A., Pshezhetsky A. V., van Karnebeek C. D.M., Lefeber D. J. (2018). Sialic acid catabolism by N-acetylneuraminate pyruvate lyase is essential for muscle function. JCI Insight 3: e122373.
Xia L., Ju T., Westmuckett A., An G., Ivanciu L., McDaniel J. M., Lupu F., Cummings R. D., McEver R. P. (2004). Defective angiogenesis and fatal embryonic hemorrhage in mice lacking core 1–derived O-glycans. The Journal of Cell Biology 164: 451-459.
Xu L., Zhang Z., Sun X., Wang J., Xu W., Shi L., Lu J., Tang J., Liu J., Su X. (2017). Glycosylation status of bone sialoprotein and its role in mineralization. Experimental Cell Research 360: 413-420.
Zhang H., Vutskits L., Calaora V., Durbec P., Kiss J. Z. (2004). A role for the polysialic acid – neural cell adhesion molecule in PDGF-induced chemotaxis of oligodendrocyte precursor cells. Journal of Cell Science 117: 93-103.
Zizioli D., Codenotti S., Benaglia G., Manzoni M., Massardi E., Fanzani A., Borsani G., Monti E. (2023). Downregulation of Zebrafish Cytosolic Sialidase Neu3.2 Affects Skeletal Muscle Development. International Journal of Molecular Sciences 24: 13578.