Int. J. Dev. Biol. 66: 199 - 209 (2022)
Special Issue: Developmental Biology in Greece
Cend1 and Neurog2 efficiently reprogram human cortical astrocytes to neural precursor cells and induced-neurons
Original Article | Published: 21 September 2021
Abstract
Direct reprogramming of glial cells into induced-neurons is a promising strategy for CNS repair after acute injury or neurodegenerative diseases. Grey matter astrocytes, which exhibit features of neural stem cells when activated, are an ideal cell source for direct neuronal conversion. The aim of the study is the investigation of the neuronal reprogramming capacity of CEND1 and/or Neurogenin-2 (NEUROG2) upon their overexpression on primary human adult cortical astrocytes. Our data indicate that adult human cortical astrocytes can be directly reprogrammed by either CEND1 or NEUROG2 to cells with differentiated neuronal morphology, exhibiting long neurites and branched processes. Exploration of gene expression dynamics along the conversion process revealed that neuronal genes are significantly up-regulated while astrocytic genes are down-regulated. Differentiated induced-neurons (iNs) exhibit either GABAergic or glutamatergic/dopaminergic identity upon CEND1 and NEUROG2 overexpression respectively. Co-expression of CEND1 and NEUROG2 in double-transduced cultures induced elevated expression levels of neural progenitor/stem genes and appearance of highly proliferative spheres with neural progenitor cell (NPC) properties in culture.
Keywords
human astrocytes, induced-neurons, CEND1, NEUROG2, trans-differentiation
Introduction
The irreversible loss of neurons is an important pathological feature of CNS injury and disease, resulting in persistent neurological disability. Subsequently, neuron regeneration in the adult mammalian brain is important for alleviation of brain injuries or neurodegenerative diseases. However, the regenerative capacity of the mammalian CNS is not only largely restricted to the two areas of the subgranular and the subventricular zones; it is also strictly limited due to the the neurons’ inability to self renew (Hashemian et al.2015; Goldman, 2016). Since the generation of induced pluripotent stem cells by Takahashi and Yamanaka (2006), cell replacement therapy using exogenous cells has attracted attention as a potential therapeutic strategy for CNS injury and disease. However, potential risks, such as tumorigenesis and the difficulty of transplanting cells into the brain, are obstacles partially hindering its wider application (Chen et al. 2015; Hu et al. 2015).
Following nerve injury, glial cells - including astrocytes, NG2 cells, and microglia - proliferate and become reactive glial cells forming the glial scar in order to protect the neighboring CNS tissue from further damage (Burda and Sofroniew, 2014). Any injury transforms astrocytes into their activated status with great self-renewal capacity and neural stem cell characteristics, which makes them more susceptible to reprogramming. Astrocytes developmentally originate from the same precursor cells as neurons and are capable of proliferating in response to brain damage, and therefore are considered to be ideal starting cells to regenerate neurons in situ (Arlotta and Berninger, 2014). Additionally, reactive astrocytes share many characteristics with NSCs capable of generating neurons, astrocytes, and oligodendrocytes in the adult brain (Huang and Tan, 2015). Thus, astrocytes are likely to be ideal starting cells for neuronal conversion.
Previous reports have shown that astrocytes have been successfully directly reprogrammed in vitro into different types of functional mature neurons using defined transcription factors, such as NEUROG2, ASCL1 and SOX2 (Berninger et al.2007; Heinrich et al. 2011). Importantly, more recent studies have proven that the in vivo resident astrocytes can be also converted directly into functional neurons (Niu et al. 2013; Guo et al. 2014). In a number of other studies, use of a single transcription factor, such as SOX2, NEUROD1 or ASCL1, in adult mammal brain and/ or spinal cord was sufficient to convert astrocytes into mature neurons (Niu et al. 2013; Guo et al. 2014; Su et al. 2014). Furthermore, it has been shown that small molecules are capable of reprogramming mouse astrocytes and human fetal astrocytes into neuronal cells (Cheng et al. 2015; Zhang et al. 2015). However, to date, only in one study were human astrocytes specifically trans-differentiated into induced glutamatergic neurons using a complicated combination of nine small molecules (Zhang et al.2015). Protocols of direct chemical reprogramming, which are still at an early stage of their implementation, are of particular direct translational interest, as they produce newborn neurons fast, avoid use of viral vectors for transgene(s) expression and offer the possibility of therapeutic use of the endogenous astrocytes of the injured brain after their in vivo modification with neurogenic molecules.
We have previously shown that forced expression of the neurogenic protein CEND1 or the proneural transcription factor NEUROG2 in both mouse cortical astrocytes and embryonic fibroblasts resulted in acquisition of induced-neuronal cells expressing glutamatergic, dopaminergic and GABAergic markers, while Cend1/NEUROG2 double-transduced mouse astrocytes and fibroblasts formed free-floating spheres exhibiting neural stem cell properties (Aravantinou-Fatorou et al., 2015). These spheres are highly proliferative, and in the absence of growth factors, they spontaneously differentiate into neurons, astrocytes and oligodendrocytes, suggesting that they have the same multipotent potential as neural stem cells of the subventricular zone (SVZ). Thus, the population of endogenous activated astrocytes, which significantly increase in number and size after injury, retain the epigenetic memory of their origin, and appear to be a cell source that can be used to produce new neurons at the site of a brain lesion. Moreover, RT-PCR arrays analysis revealed that components of the beta-Catenin/ Wnt-signaling pathway to be up-regulated in CEND1/NEUROG2-double transduced cultures are probably responsible for the formation of astrospheres exhibiting NSCs characteristics. Additionally, we have shown that Cend1 acts downstream and is regulated by NEUROG2, while at the same time it enhances the expression of NEUROG2 in reprogrammed astrocytes, indicating the existence of a cross-activating feedback loop between the two molecules (Aravantinou-Fatorou et al. 2015).
Despite the development of protocols for reprogramming astrocytes and other types of mouse glial cells, and the creation of transgenic animal models that resemble human diseases or brain damage, it is still necessary and imperative to study reprogramming and differentiation of human astrocytes, as critical differences exist between human and rodent astrocytes. Human astrocytes in the neocortex are 27-fold larger and have 10 times more processes, which are 2.6-fold longer compared with those in rodents (Robertson, 2014). Additionally, astrocytes of rodents cover 20,000-120,000 synapses, while human astrocytes cover 270,000-200,0000 synapses (Oberheim et al. 2012). Functionally, human astrocytes outcompete their rodent counterparts, as presented in an elegant study in which human glia progenitors were transplanted into mouse forebrains, showing that human glia progenitor cells migrated, integrated and matured to astrocytes, resulting in a mouse with improved long-term potentiation (LTP) and learning compared to mice in which murine progenitors were grafted (Han et al. 2013). Thus, morphological and functional variations present in human astrocytes make it essential to study their properties and their ability to get reprogrammed to neurons.
Thus, the aim of this study is to investigate whether the two neurogenic molecules, CEND1 and NEUROG2, are also capable of reprogramming adult human astrocytes to neural progenitor cells and neurons, in particular of GABAergic, dopaminergic and glutamatergic subtypes, as they do in mice. This question brings this line of research one step closer to clinical approaches through future translational exploitation of our experimental results.
Results
Characterization of cultured human adult astrocytes
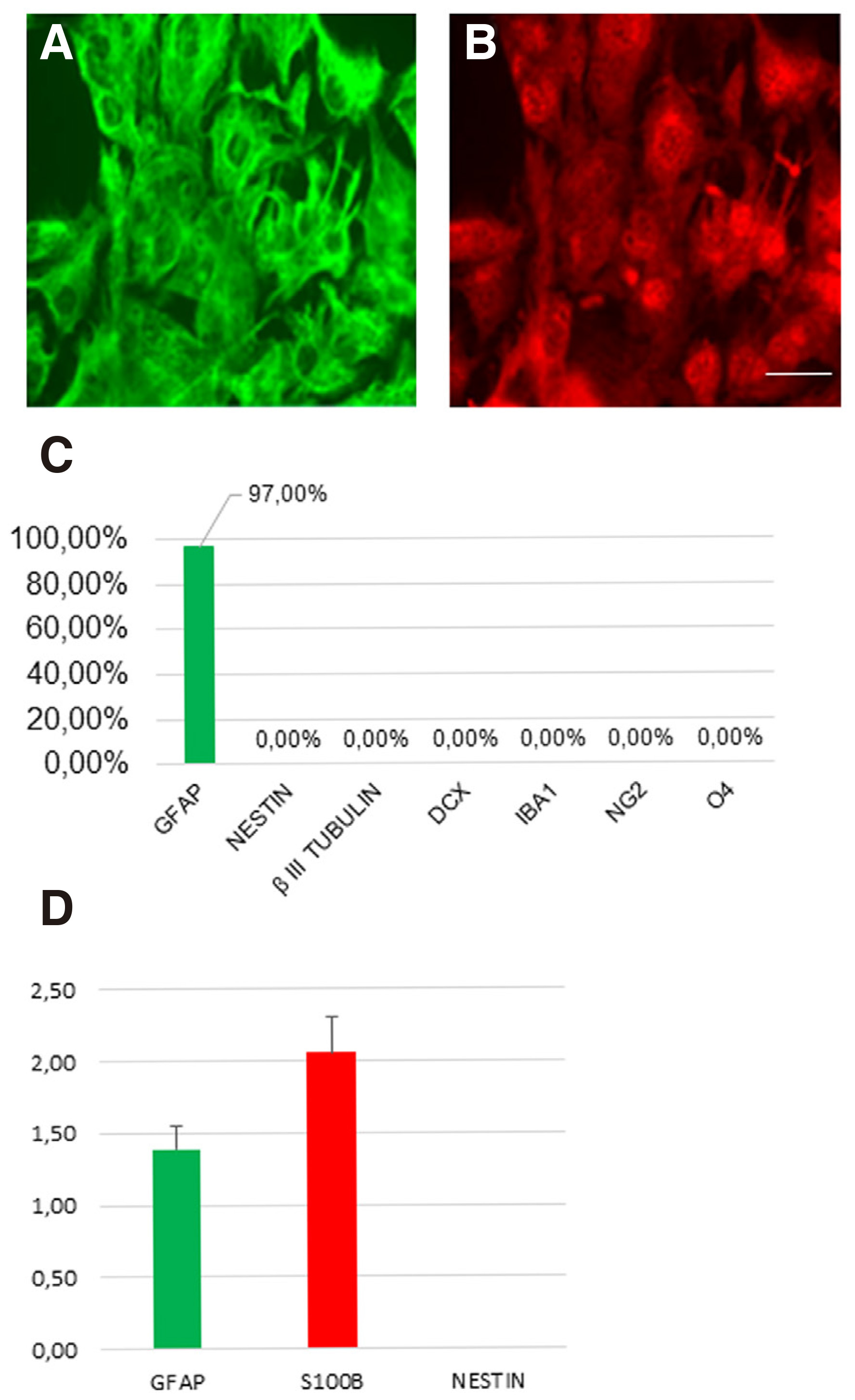
Human cerebral cortex astrocytes (HA#1800) were purchased from ScienCell (San Diego, CA). To check the possibility of presence of neural progenitor cells (NPCs) in the astrocytic culture, we cultured the cells in the presence of 10% fetal bovine serum (FBS). After more than one month of culture in basic astrocytic medium, the cells are unable to form neurospheres or any neuronal cells, and about 97% of the cells express the astrocytic markers Glial Fibrillary Astrocytic protein (GFAP) and S100B (Fig. 1 A-B). In addition, the cultured cells are negative for the neuronal markers βIII-TUBULIN, doublecortin (DCX), radial glial marker NESTIN, microglial marker IBA1, glial marker NG2, or oligodendrocyte marker O4 (Fig. 1C). These results collectively suggest that the cultured cells are pure astrocytes without detectable contamination of NPCs, neuronal cells, or other glial cells. Besides, the real time RT-PCR results for GFAP mRNA expression levels confirm that we have a homogeneous population of primary astrocytic cells with stable functional properties as the basis for our cell reprogramming experiments (Fig. 1D). More specifically, qRT-PCR analysis revealed that the mRNA levels of S100b are higher compared with GFAP, in accordance with previous reports showing higher expression of S100b in grey matter astrocytes and GFAP in white matter astrocytes, respectively (Rusnakova et al.2013; Ben Haim and Rowitch, 2016).
Fig. 1. Characterization of human adult astrocytes in vitro.
(A-B) Human astrocytes stained for GFAP marker (green) and S100β marker (red). (C) The cultured astrocytes are negative for the markers βIII-tubulin, DCX, IBA1, NG2 and O4. (D) Real-time PCR validation of the expression of representative astrocyte-enriched genes. (mean ± SEM, n = three independent experiments). Scale bar: 40μm.
Direct reprogramming of human cortical astrocytes to induced neurons with subtype-specific identity upon CEND1 and NEUROG2 overexpression
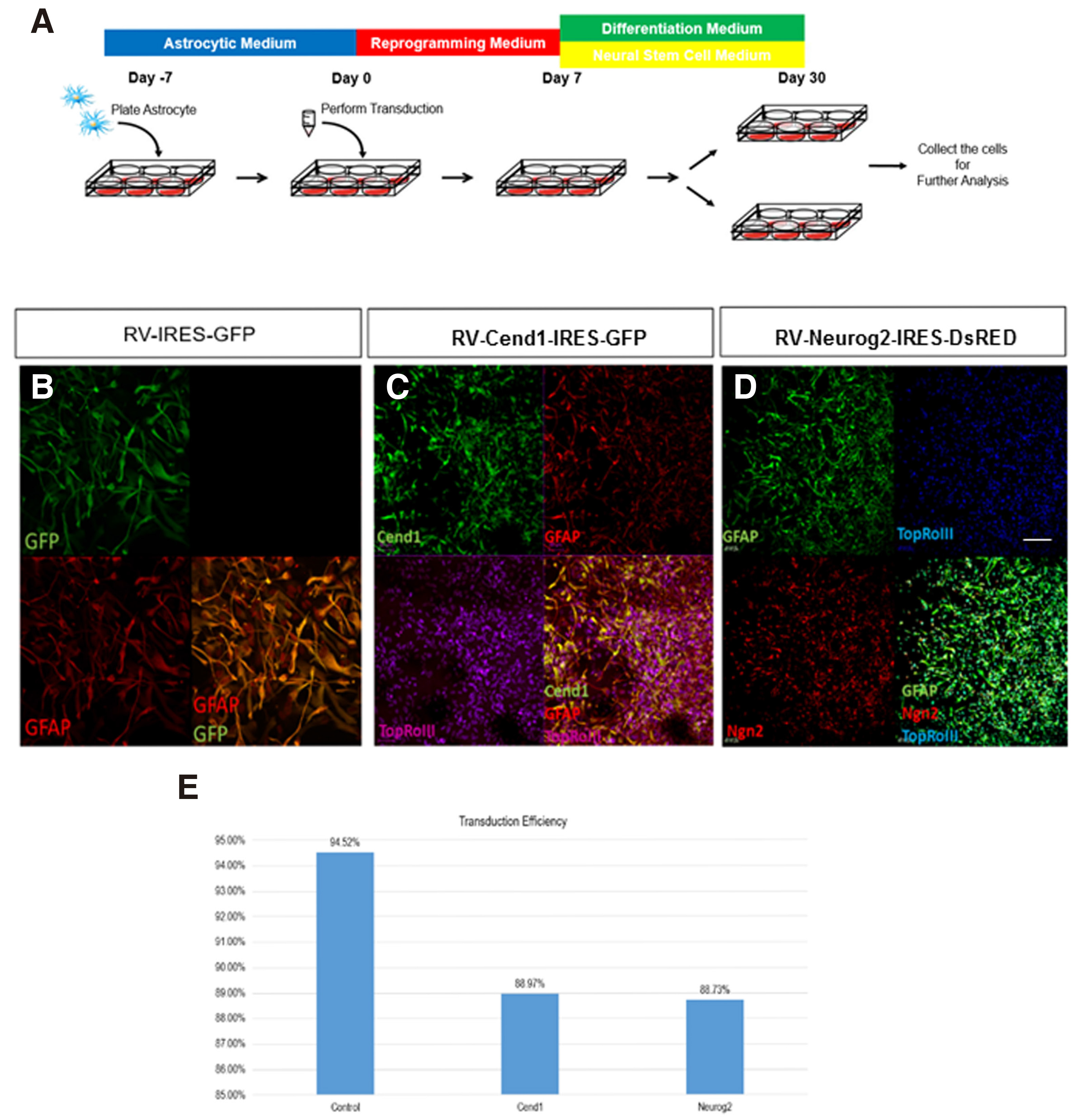
The aim of the study is the investigation of the neuronal reprogramming capacity of the two neurogenic molecules CEND1 and NEUROG2, used either alone or in combination, upon their overexpression on human adult primary cortical astrocytes. CEND1 and NEUROG2 are expressed under the control of chicken beta-actin promoter (pCAG) - along with expression of the fluorescent proteins GFP and DsRed, respectively - using the pCAG-Cend1-IRES-GFP and pCAG-Neurog2-IRES-DsRed retroviral vectors (Aravantinou-Fatorou et al.2015). The small molecules, Forskolin and Valproic acid, both frequently used in direct neuronal reprogramming or differentiation protocols (Jung et al. 2008; Liu et al.2013; Hu et al. 2015) have also been applied in the reprogramming cocktail. To induce direct reprogramming of human astrocytes, we have developed a multistep culture protocol summarized in Fig. 2A. Briefly, astrocytes are transduced and cultured under reprogramming media until day 7, reprogrammed cells are then re-plated and cultured in neuronal differentiation medium or neural stem cell medium. Immunofluorescence labelling for GFP, CEND1 and NEUROG2 at the early 72h time-point revealed that in all cases transduction efficiency is very high, varying between 88-95% of cells in culture (Fig. 2 B-E).
Fig. 2. Astrocytes reprogramming upon forced expression of CEND1 and/or NEUROG2.
(A) Schematic drawing of the protocol used for astrocytes reprogramming. (B-D) Confocal images showing the high retroviral transduction efficiency in control (B), CEND1 (C) and (D) NEUROG2-transduced cultures, indicated by GFP (green) and DsRED (red) expression, respectively. (E) Quantification of transduction efficiency from five independent reprogramming experiments. Scale bar: 40μm.
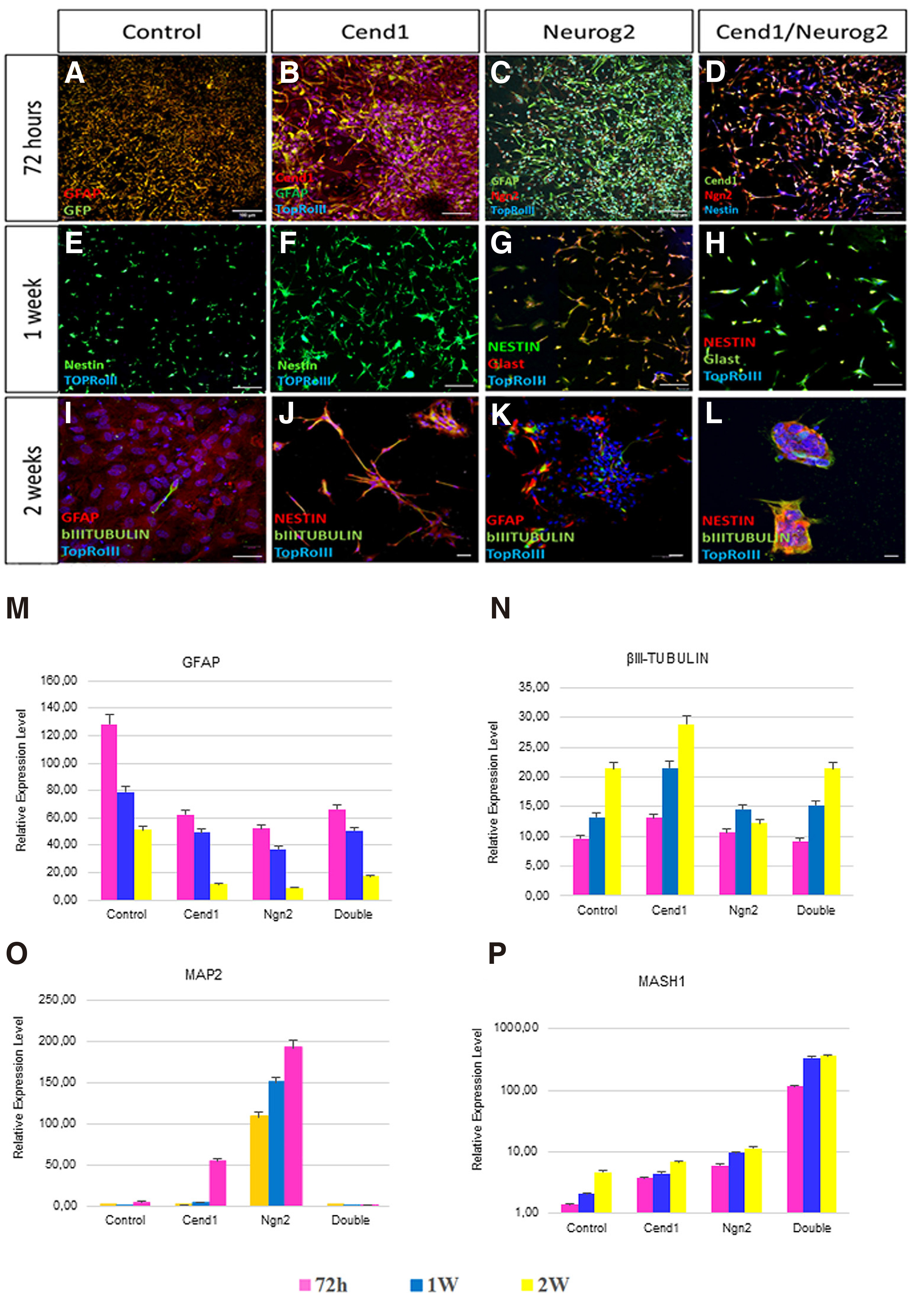
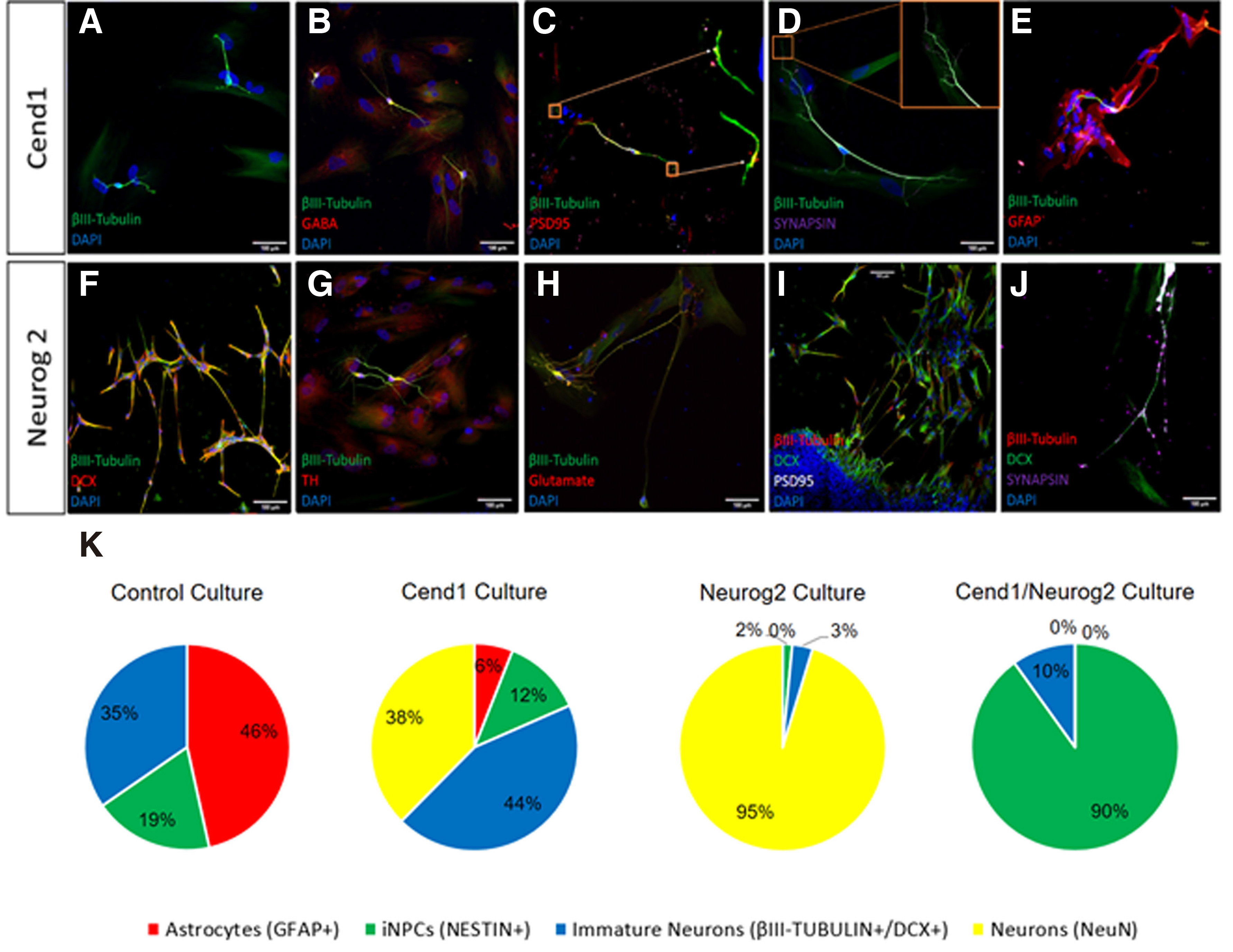
72 hours after transduction, the vast majority of cells in control GFP-virus-transduced cultures are positive for Glial fibrillary acidic protein (GFAP) (Fig. 2B, 3A). Overexpression of either CEND1 or NEUROG2 results in a decrease in the number of GFAP+ astrocytes (Figs. 2 C-D, 3 B-C). After 7 days in reprogramming medium, molecular phenotype analysis reveals that a significant number of NESTIN+ cells are present in control cultures, not exposed to either neurogenic molecule (Figs. 3E, 4B). Additionally, very few of them differentiate into βΙΙΙ tubulin+ cells that never mature any further (Fig. 3I), possibly indicating that the culture conditions can activate a very low level of initial neurogenic reprogramming of human astrocytes. Upon CEND1 overexpression, the percentage of NESTIN+ cells is much higher compared with the control culture (Figs. 3F, 4D); while, as soon as cells are transferred to differentiation media, the population of NESTIN+ neural progenitors in CEND1-transduced cultures decrease and βIII-TUBULIN+ neurons appear in culture (Figs. 3J, 4 E-F). This reduction of NESTIN+ and subsequent increase of βIII-TUBULIN+ (Fig. 3 G-K) and more mature NeuN+ neurons (Fig. 4 H-I) is also obvious in NEUROG2 single-transduced cells. At the same time, in NEUROG2 cultures, newborn neurons, besides their higher maturation level, also express subtype-specific neuronal markers, such as Tyrosine hydroxylase (TH) , for the first time (data not shown).In double-transduced cultures, a significant percentage of NESTIN+ and Glast+ progenitors appear (Fig. 3 D,H,L; Fig. 4 J-L), amounting to 90% of the cell population after two weeks in culture, indicating that combined CEND1 and NEUROG2 forced expression induces a cell fate choice toward proliferating multipotent neural precursors, whereas higher expression of these molecules in single-transduced cells drives neuronal differentiation.
Fig. 3. CEND1 and/or NEUROG2 overexpression drives astrocytes towards neural progenitor and immature neuronal phenotype.
(A-D) GFAP expression pattern starts to change among the different groups even 72 hours following viral transduction. (E-H) 1 week after transduction, NESTIN and Glast are expressed by the neural progenitor cells that emerge upon CEND1 and NEUROG2 overexpression. (I-K) The reduction in expression of NESTIN+ cells and increase in appearance of bIII-TUBULIN+ neuronal cells with elongated axonal morphology is obvious two weeks after transduction. (L) In double transduced cultures, spherical structures (astrospheres) are formed, comprised of NESTIN+ and bIII-TUBULIN+ cells. (M-P) Real-time RT-PCR analysis of the expression of representative astrocyte- (M), neuron- (N-O), and NPC-enriched (P) genes. (mean ± SEM, n = three independent experiments).Scale bar: 40μm.
Exploration of gene expression dynamics throughout the entire conversion process reveals that neuronal genes (MAP2, and βIII-TUBULIN) are significantly up-regulated (Fig. 3 N-O), while at the same time, the astrocytic gene Glial fibrillary acidic protein (GFAP) is down-regulated (Fig. 3M), indicating that the induced-neuronal cultures are proceeding to a neuronal state. On the other hand, in double-transduced cultures that trans-differentiate primarily to a NPC-like phenotype, the levels of the late neuronal differentiation gene MAP2 are low (Fig. 3O), while the relative expression of bHLH transcription factor MASH1 is high in neural progenitors (Fig. 3P).
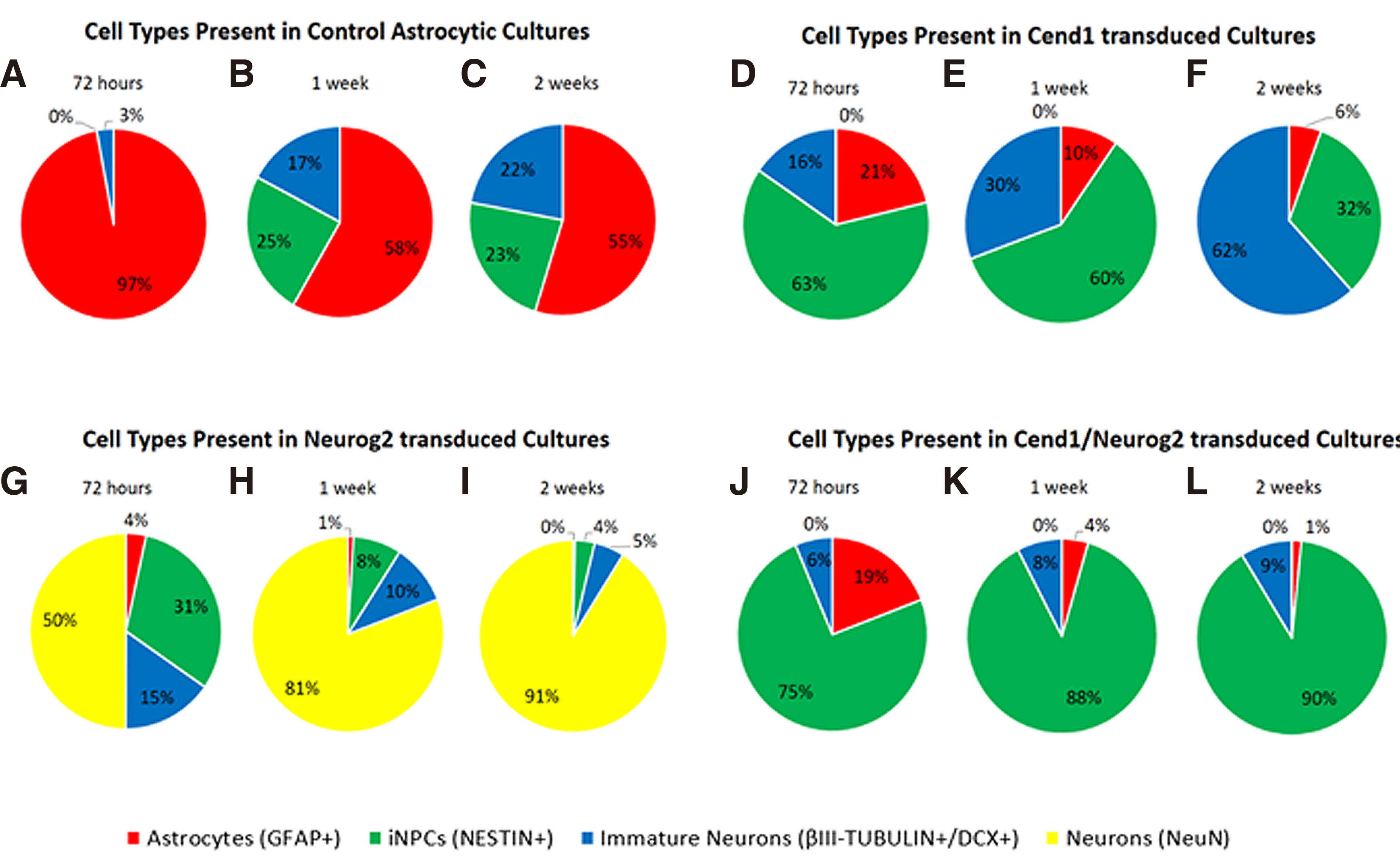
Fig. 4. Molecular phenotype of cells during the first 2 weeks of astrocytic reprogramming.
(A–H) Quantification of at least three independent experiments showing different cell types present in control astrocytic cultures (A-B), CEND1 (C-D), NEUROG2 (E-F) and double-transduced (G-H) cultures. The most profound changes occur in double transduced cultures, where 87.94% of the cells become NESTIN+ neural progenitor cells (green). By contrast, in NEUROG2-transduced cultures, the vast majority of the reprogrammed cells are NeuN+ neurons (yellow,) even from 72 hours post-transduction. Finally, in CEND1-transduced cultures, most of the cells are either NESTIN+ neural progenitor cells (green) or β-III TUBULIN+ neural progenitors (blue). GFAP-expressing cells appear red.
Generation of induced-neurons (iNs) from human adult astrocytes is achieved after one month in culture. At this time point, a significant percentage of reprogrammed astrocytes exhibit a highly differentiated neuronal morphology, with long neurites and multiple and complex processes emanating from their small cell somas. Quantification of induced-neurons expressing β-III TUBULIN+ or NeuN+ revealed that they amounted to 82% and 98% of total cells in culture following Cend1 or Neurog2 overexpression, respectively (Fig. 5K). In addition to their differentiated branched morphology and neuronal markersβ-III TUBULIN, doublecortin and NeuN expression (Fig. 5 A,E,F,J), reprogrammed astrocytes started expressing the neuronal subtype-specific markers GABA (Fig. 5B), Tyrosine Hydroxylase (TH) (Fig. 5G), and GLUTAMATE (Fig. 5H), depending on the neurogenic factor being force-expressed. In particular, 29% ± 3.4% of the β-III TUBULIN+ neurons are GABA+ upon CEND1 overexpression, while in NEUROG2- transduced cultures, 34% ± 5.1% of β-III TUBULIN+ neurons are TH+ and 18% ± 7.1% of β-III TUBULIN+ neurons are GLUTAMATE+. Moreover, the pre- and post-synaptic markers SYNAPSIN 1 and PSD95 appear (Fig. 5 C-D, I-J), indicating further maturation of the induced neurons. Quantification of the total neurite length in the three different neuronal subtypes produced (GABA+, TH+, GLUTAMATE+) indicates that average total neurite length is 39 ± 0.4 μm in the CEND1-overexpressing for GABA+ neurons and 53 ± 0.5 μm in the NEUROG2-overexpressing for TH+ neurons, whereas NEUROG2-overexpressing glutamatergic neurons exhibit the highest neurite length of 110 ± 0.7 μm. By contrast, no significant morphological change is observed in the control group, where the viruses and the small molecules are not added (data not shown). However, in the control cultures, cultured in reprogramming medium, we observe a spontaneous neuronal differentiation phenomenon, where few immature βIII-TUBULIN+ neuronal cells appear (Fig. 5A). It is worth mentioning that the remaining non-converted astrocytes form a neuron-astrocyte co-culture condition, with the iNs migrating onto the surface of astrocytes, and the astrocytes serving as a feeder layer to promote neuronal survival and maturation (Fig. 5E) (Wu et al. 2013).
Fig. 5. Reprogrammed astrocytes cultured for one month exhibit differentiated neuronal morphology and molecular phenotype.
(A-D) In CEND1-transduced cultures, differentiated β-III TUBULIN+ subtype-specific neuronal cells, exhibiting GABAergic phenotype and synaptic proteins expression, are present. (E) A more mature morphology is observed mostly in β-III TUBULIN+ neurons lying on top of the remaining astrocytes. (F-J) Upon NEUROG2 overexpression, a significant population of cells are highly branched DCX+ and βIII-TUBULIN+ neurons expressing subtype-specific markers, GLUTAMATE and Tyrosine Hydroxylase (TH) and synaptic markers PSD95 and Synapsin-1. (K-N) Percentages of cellular composition one month following transgene(s) overexpression reveal that: (K) In control cultures, astrocytes (red), immature neurons (blue) and neural stem/progenitor cells (green) are present at percentages of 46.54%, 34.63% and 18.84%, respectively. (L) In CEND1-transduced cultures, 37.58% of cells are DCX+ immature neurons (yellow), 44% β-III TUBULIN+ neurons (blue), 12.43% NESTIN+ neural stem cells (green) and 6% GFAP+ astrocytes (red) appear. (M) Importantly, 95.3% of the astrocytes transduced with the NEUROG2 virus are DCX+ and /or NeuN+ neurons, followed by 3.21% of β-III TUBULIN+ immature neurons, 1.47% of NESTIN+ NPCs and 0.03% GFAP+ astrocytes. (N) Finally, the majority of the remaining cells in double transduced cultures contain a pure NPC population (90.07% NESTIN+, green).Scale bar: 40μm.
Long-term time-lapse experiments reveal two modes of astrocytic reprogramming to induced-neurons
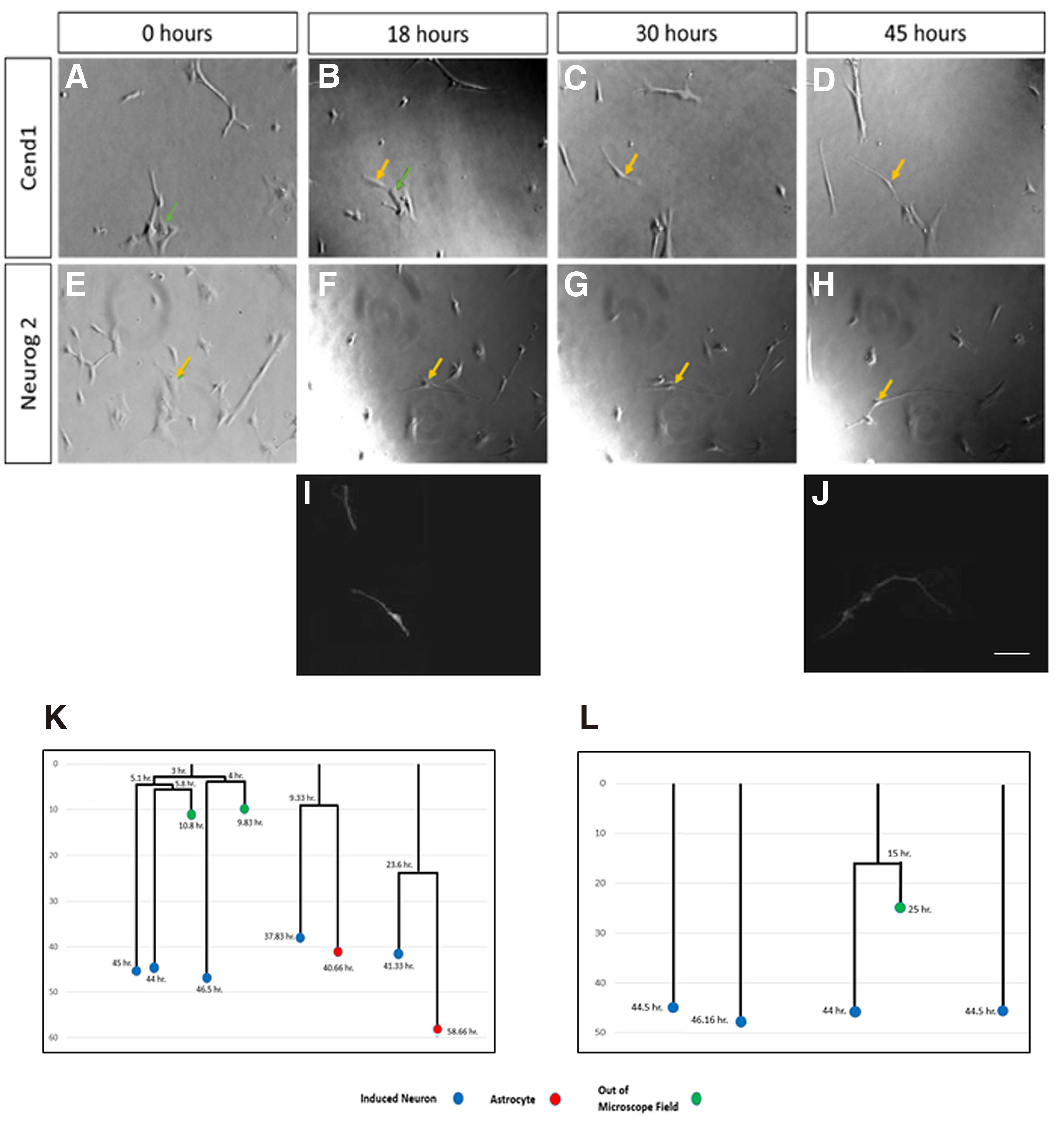
In order to investigate whether cell division is required for cell fate conversion to occur, or astrocytes directly trans-differentiate to post-mitotic neurons following forced expression of CEND1 or NEUROG2, we performed continuous live-cell imaging for up to five days, using an Olympus IX81 time-lapse imaging system and Cell Profiler software to track transduced cells’ lineage trees. Cell lineage analysis revealed that the majority of CEND1-overexpressing astrocytes passed through one to two asymmetrical divisions, before neuronal trans-differentiation (Fig. 6 A-D,I,K; Video S1), just as previously described during reprogramming of mouse astrocytes (Aravantinou-Fatorou et al. 2015). By contrast and in accordance with our previous observations with mouse astrocytes (Heinrich et al. 2010; Aravantinou-Fatorou et al. 2015), newborn neurons derived from NEUROG2-transduced astrocytes are rarely produced in a proliferative manner (Fig. 6 E-H,J,L; Video S2). Furthermore, in both cases, during the first days astrocytes are very motile, while as soon as neuronal trans-differentiation occurs, their motility becomes more limited, and by the time they acquire neuronal identity, their cell bodies stop moving and only their processes head in different directions, scavenging the environment (Videos S1, S2).
Fig. 6. Long-term live-cell imaging upon CEND1 or NEUROG2 overexpression.
Astrocytic cultures transduced with the CEND1-IRES-GFP (A-D and I) or NEUROG2- IRES-DsRed (E-H and J) retroviruses trans-differentiated, after passing through one or two divisions (K) or directly (L), giving rise to β-III TUBULIN+ neurons (I-J). Snapshots of the following time points during five days live cell imaging: 0 hr (A, E), 18 hr (B, F), 30 hr (C, G), 45 hr (D, H). (K) 80% of the divisions in CEND1-transduced cultures are asymmetric, giving rise to one neuron and one astrocyte (lineage trees K, video S1), while in NEUROG2-transduced cultures (L), the vast majority of astrocytes directly form neurons at a very short time interval (lineage trees L, video S2). Blue circles refer to neurons, red circles to astrocytes, and green circles to the cells lost from field of view. Scale bars, 40 mm. (I-L) Cell tracking and lineage tree drawings are correlated with bIII-TUBULIN+ neurons produced.
CEND1 /NEUROG2-double transduced astrocytic cultures form multipotent astrospheres
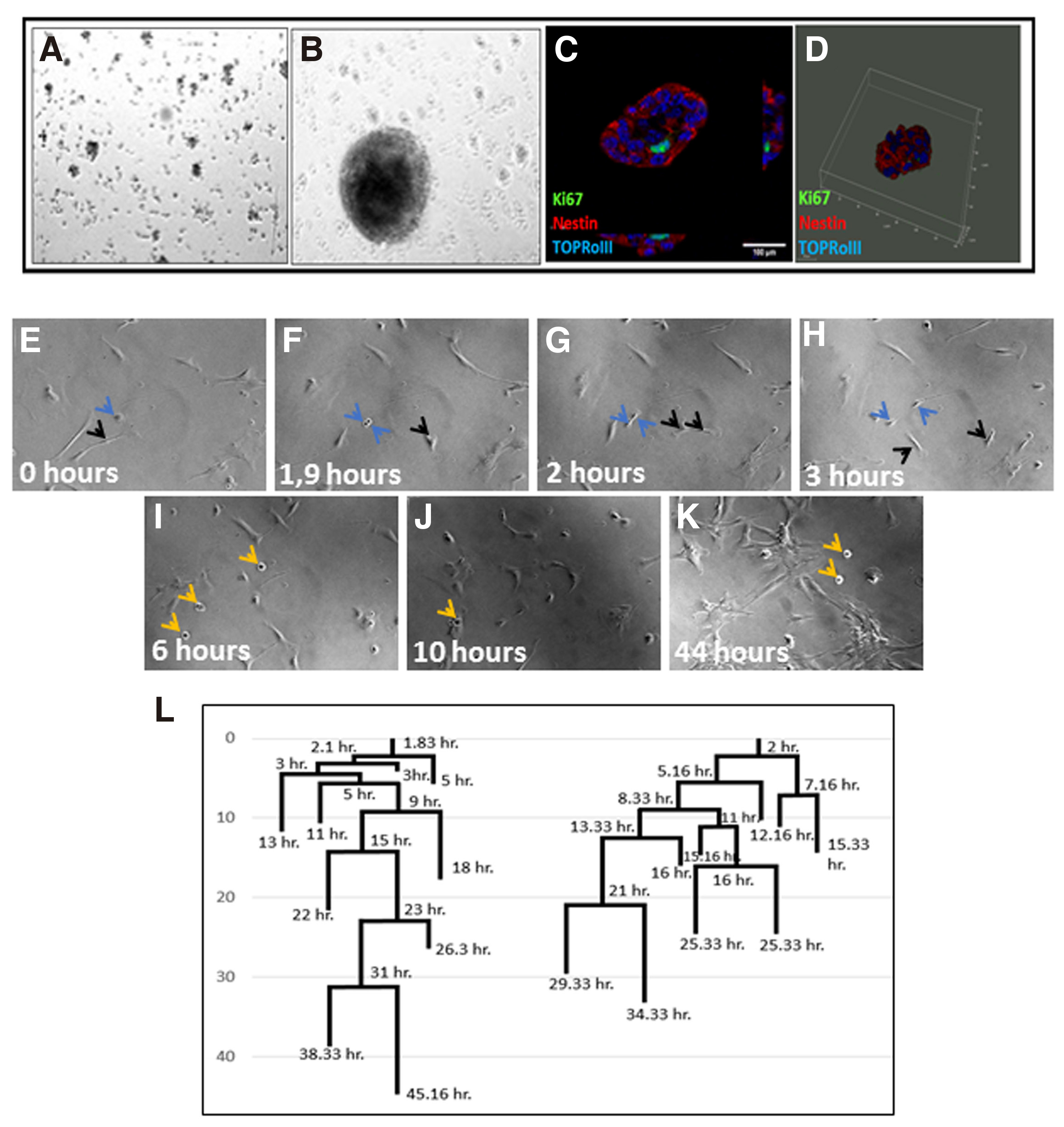
In double-transduced cultures, colonies of small round cells form highly proliferative three-dimensional spheres attached to the plate, detected around 60 hours after transduction, as revealed by live cell imaging experiments (Fig. 7 A-D). These clones, which amount to approximately 90% of the whole cell population (Fig. 5K), constitute a transient population that cannot survive for more than 72 hours when cultured in astrocyte medium and is composed of cells with a mean diameter of 22.1-24.8 μm. Live-cell imaging starting 12 hours after transduction, combined with linage tracing, enabled us to visualize the exact time of the first divisions resulting in sphere formations that take place during the first 20 hours following CEND1/NEUROG2 double transduction (Fig. 7 E-L; Video S3).
Fig. 7. Astrospheres’ formation upon CEND1/NEUROG2 overexpression.
(A) NPC-like colonies 60h after CEND1/NEUROG2-double transduction. (B) A mature astrosphere grown after 1 week in culture. (C) All cells express the neural precursors marker NESTIN. (D) 3D demonstration of a sphere attached to the plate. (E-L) Long time live cell imaging experiment showing the process of the formation of the free-floating astrospheres. (F-H) Astrocytes start proliferating fast. The blue arrows point to distinct divisions on this area. As soon as astrospheres are formed, they detach from the bottom of the well and start to float (I-K, black arrows, video S3). (l) The first divisions resulting in sphere formations take place during the first 20 hours following CEND1 / NEUROG2 double transduction.
Discussion
Direct lineage reprogramming of astrocytes to induced-neurons (iNs) provides a valuable cell –based approach for regenerative medicine, drug discovery and disease modeling. Because of the ubiquitous distribution of astrocytes in the CNS, their close lineage to neurons (Chandrasekaran et al. 2016; Zheng et al. 2018) and their endogenous tendency to proliferate following brain trauma and in some cases convert into neural precursors and functional neurons (Torper et al. 2013; Magnusson et al. 2020), astrocytes have been considered an ideal candidate for neurogenic reprogramming. In the present study, we show that two neurogenic factors, Cend1 and Neurogenin2, in combination with the small molecules Forskolin and valproic acid (VPA) , can trans-differentiate human primary adult cortical astrocytes into (a) differentiated neurons possessing GABA+, TH+, or GLUT+ subtype specificity and (b) multipotent NPCs. Use of the small molecule VPA has been selected for enhancing reprogramming, as it has been reported to promote neurogenesis and neuronal maturation (Hsieh et al. 2004; Niu et al. 2013). It is also reported that VPA, as a histone deacetylase inhibitor, can increase reprogramming efficiency and even reduce the number of factors required for reprogramming (Biswas and Jiang, 2016).The second chemical molecule used, forskolin, has been identified to reduce lipid peroxidation and promote neuronal conversion efficiency (Liu et al. 2013; Gascón et al. 2016), and was included in the reprogramming cocktail enhancing cells: morphological maturation (Gao et al. 2017).
Currently, mouse astrocytes have been successfully reprogrammed into different types of functional mature neurons using defined transcription factors in vitro (Heinrich et al. 2011; Péron and Berninger 2015), while the protocols for differentiating induced pluripotent stem cells (iPS) to mature, subtype-specific neurons are continuously improving (Lundin et al. 2018). The main goals in the direct astrocytic reprogramming field are to simplify and shorten the process, standardize the culture conditions, and validate induced-neurons’ functional properties not only in vitro but also in vivo (Zheng et al. 2018). Transcription factors (TFs) are considered as the major determinants of specific cell lineages and lineage conversions (Mertens et al. 2015), while small molecules enable TFs to induce neuronal conversion more efficiently (Gascón et al.2016; Smith et al. 2016). Our previous study (Aravantinou-Fatorou et al. 2015), in accordance with other reports, further supports that, ectopic expression of neurogenic factors, in some cases along with certain small chemical molecules, could activate key neuronal Transcription Factors in fibroblasts or astrocytes and induce their reprogramming to mature neuronal cells (Cheng et al.2015; Hu et al. 2015; Zhang et al. 2015).
CEND1 is a neuronal‐lineage specific modulator involved in synchronization of cell cycle exit and differentiation of neuronal precursors. It is expressed throughout the neuronal lineage, from neural stem/progenitor cells to mature neurons, and is associated with the dynamics of neuron‐generating divisions of both embryonic and postnatal NPCs (Politis et al.2007; Katsimpardi et al. 2008; Gaitanou et al. 2019) and, as shown, here of primary astrocytes in vitro. Achaetescute homolog 1 (ASCL1) and NEUROG2 are the prominent pro-neural factors responsible for neuronal identity specification in the nervous system. Our previous data indicate that CEND1 participates in bHLH proneural genes pathway(s) and is activated by bHLH factors, such as NEUROG1/2 and MASH1 in neuronal precursors (Papadodima et al. 2005; Politis et al. 2007; Katsimpardi et al. 2008). While Neurog2 is expressed in dorsal progenitors and instructs them to generate glutamatergic neurons (Colasante et al. 2019), CEND1 has a wider neuronal distribution, being highly enriched in GABAergic striatal neurons and motor neurons of the spinal cord. Along these lines, transplantation of neural stem /precursor cells over-expressing CEND11 in the injured mouse cortex leads to their differentiation to GABAegic neurons (Makri et al. 2010). On the other hand, in vivo CEND1-overexpresssion in the early chick neural tube drives neural precursors towards both ventral and dorsal neuronal identities (Politis et al. 2007). Thus, unlike NEUROG2, CEND1 seems to exhibit a general—not cell-type-specific— potential in conferring neuronal identity, a fact that explains the different neuronal phenotypes present upon CEND1 or NEUROG2 overexpression.
Our new results using human adult astrocytes demonstrate that: (1) the neurogenic molecule CEND1 can reprogram human astrocytes towards subtype-specific GABAergic neurons, while NEUROG2 drives them towards acquisition of glutamatergic and dopaminergic neuronal identity and (2) simultaneous overexpression of CEND1 and NEUROG2 results in activation of the multipotent characteristics of astrocytes, transforming them into free floating astrospheres of high proliferative capacity. Molecular phenotype analysis of single-transduced cultures also indicates that the induced-neurons produced are positive not only for neuronal markers, but also for synaptic proteins, supporting their functional maturation. On the other hand, astro-derived spheres are self-renewing, presenting specific NPCs-like morphology and molecular marker expression, and can be propagated for more than ten passages, exhibiting proliferation and differentiation characteristics similar to human neural progenitor cells. More specifically, withdrawal of epidermal growth factor 2 (EGF2) and basic fibroblast growth factor (bFGF) from the astrosphere cultures leads to production of βIII TUBULIN+ neurons, GFAP+ astrocytes and O4+ oligodendrocytes, proving their multipotency properties. Additionally, our long term live-cell imaging experiments have shown that human astrocytic trans-differentiation to neurons is not always direct; depending on the reprogramming factor being force-expressed, human astrocytes may undergo a limited number of divisions before giving rise to post-mitotic neurons. This is the case for CEND1, which unlike NEUROG2, allows a couple of asymmetric divisions prior to neuronal reprogramming. In contrast, we observed a de-differentiation of astrocytes to an NPC state upon simultaneous overexpression of both proteins. Our outcomes on human cells are in line with our previous results on mouse primary astrocytes and embryonic fibroblasts (Aravantinou-Fatorou et al. 2015).
From a translational point of view, it is significant that CEND1 and NEUROG2 share the potential to be used in gene therapy approaches to enhance the intrinsic neuronal reprogramming capacity of endogenous astrocytes, or in cell therapy approaches involving transplantation of human reprogrammed cells into the injured brain to enhance functional recovery. Therefore, the synergetic effect and underlying mechanism of action of the two molecules in neuronal conversion in a human setting need to be further investigated. Moreover, whether the small molecules used in this study can also enhance the in vivo reprogramming capacity of resident astrocytes into neuronal cells and could therefore be used in reprogramming cocktails against neurological diseases or brain injuries is still unknown. Overall, this research work enriches our knowledge of direct neuronal differentiation mechanisms. Moreover, the production of functional neuronal subtypes by human astrocytes provides regenerative medicine with new approaches for the treatment of brain trauma and neurodegenerative diseases that can be used for therapeutic purposes in the future.
Materials and Methods
Direct neurogenic reprogramming of human adult astrocytes
Human primary astrocytes derived from adult cortex (HA#1800, Science Cell san Diego) were seeded on 0.015mM poly-L-lysine (sigma) pre-treated coverslips in human astrocyte growth medium (DMEM high glucose/F12, N2, B27, NEAA, bFGF, 10% FBS, Ascorbic Acid, Glutamax). The cells were seeded at a density of 50,000 cells per square centimeter. The following day, viral transduction was performed using the retroviruses RV-IRES-GFP, RV-IRES-DSRED, RV-CEND1-IRES-GFP, RV-NEUROG2-IRES-DSRED. Two days after the seeding, the cells should be over 90% confluent. At this time point, the growth medium was replaced by the reprogramming medium, which was DMEM high glucose/Neurobasal (Gibco) supplemented with 1x B27 (Gibco), 1x N2 (Gibco), 20 ng/ml BDNF (Peprotech), 20 ng/ml IGF (Peprotech), 0.2 μM ascorbic acid (sigma), 100 μM dibutyryl-cAMP (Sigma), and the following small molecules: 0.5 mM VPA (Calbiochem) and 10 μM forskolin (Cayman). The reprogramming medium containing small molecules was changed every two days. At week 2, the culture medium was replaced by the differentiation medium, containing Neurobasal medium, 1x B27, 1x N2, 20 ng/ml BDNF, 20 ng/ml GDNF, 20 ng/ml IGF, 0.2 μM ascorbic acid, 100 μM cAMP and 1 μg/ml laminin.
Neurosphere assay
Neurosphere formation was achieved after the double transduction with the retroviruses RV-CEND1-IRES-GFP and RV-NEUROG2-IRES-DSRED on human adult astrocytes. The culture medium for neurosphere assay was DMEM/F12 supplemented with 1x B27 (Gibco), 20 ng/ml EGF2, and 20 ng/ml bFGF. The NSC medium was changed every other day.
Immunofluorescence staining
Immunostaining of cells was performed as previously reported (Katsimpardi et al. 2008). Briefly, after cells cultured on coverslips were washed with 1x PBS for 3 times, 4% paraformaldehyde (PFA) was used to fix the cells for 20 minutes at room temperature. After washing with 1x PBS three times, blocking buffer (1% BSA and 0.5% Triton X-100 in PBS) was used for 1 hour at room temperature. The cells were then incubated with primary antibodies at 4 ℃ overnight and, finally, with fluorescent probe-conjugated secondary antibodies for 1hour at room temperature. DAPI (Beyotime) or TopRoIII (Thermo Scientific) were used to stain Nuclei at room temperature at the same time with the secondary antibodies.
The primary antibodies were used as: DCX (1:200, Santa Cruz, Cat. #sc-8066), b-III TUBULIN (1:500, Covance, Cat. #MMS435P), MAP2 (1:500, Millipore, Cat. #AB5622, #MAB3418), NEUN (1:500, Millipore, Cat. #ABN78, #MAB377), SYN1 (1:500, Millipore, Cat. #AB1543), VGLUT1 (1:500, Synaptic system, Cat. #135302), TH (1:200, Chemicon, AB152), NESTIN (1:1000, Millipore, Cat. #MAB5326), SOX2 (1:50, R&D, Cat. #AF2018), GFAP (1:1000, DAKO, Cat. #Z033401), PSD95, clone K28/43 (Millipore). The following secondary antibodies were used at 1:1000 dilution: Donkey-anti-goat-Alexa 488 (Molecular Probes, Cat. #A11055), Donkey-anti-mouse-Alexa 488 (Molecular Probes, Cat. #A21202), Donkey-anti-Rabbit-Alexa 488 (Molecular Probes, Cat. #A21206), Donkey-anti-goat-cy3 (Jackson Immuno Research, Cat. #705-165-147), Donkey-anti-rabbit-cy3 (Jackson Immuno Research, Cat. #711-165-152), Donkey-anti-mouse-cy3 (Jackson Immuno Research, Cat. #715-165-150), Donkey-anti-rabbit-Alexa 647 (Molecular Probes, Cat. #A31573), Donkey anti- mouse- Alexa 647 (Molecular Probes, Cat. #A31571).
Quantification of conversion efficiency and neuronal purity
Briefly, 10-20 view fields were randomly selected for each sample by Confocal SP8 Leica microscope at each time point. The number of neuronal cells was counted on DCX-positive cells with typical neuronal morphology. The conversion efficiency was calculated by the ratio of the number of neuronal cells to that of initial cells seeded in each field. Neuronal purity was calculated by the ratio of the number of neuronal cells to the total cell number indicated by DAPI. The percentage indicated in characterizing human adult astrocytes or neuronal subtype was calculated similarly. Quantitative data were represented as average ± SEM of three independent experiments.
Time-lapse microscopy
Time-lapse microscopy (Costa et al. 2011; Ortega et al.2011) was performed at 370 C and 7% CO2. Phase contrast images were acquired every 5 min for 4 days.
Quantitative real-time RT-PCR
The total RNA of indicated cell samples were isolated with Trizol (Sigma) following manufacturer’s instructions. Isolated RNA was used for reverse transcription with random hexamers and Superscript II transcriptase (Invitrogen) according to manufacturer’s instructions. Quantitative real-time PCR was conducted with primers and SYBR Green qPCR Master Mix (4472908, Invitrogen) in Light Cycler® 480 PCR machine. The relative expression levels were normalized to the internal control (GAPDH). Primers used are listed in Table 1.
Table 1
List of primers’ sequence
| Oligo Name | Sense/Antisense | cDNA | Sequence (5’-3’) |
| GFAP F | Sense | Genomic | GGTTGAGAGGGACAATCTGG |
| GFAP R | antisense | Genomic | GGGTGGCTTCATCTGCTTC |
| TUJ1 F | Sense | genomic | CATTCTGGTGGACCTGGAAC |
| TUJ1 R | antisense | genomic | CCTCCGTGTAGTGACCCTTG |
| MAP2 F | Sense | genomic | GAGAATGGGATCAACGGAGA |
| MAP2 R | antisense | genomic | CTGCTACAGCCTCAGCAGTG |
| MASH1 F | Sense | genomic | AAGAGCAACTGGGACCTGAGTCAA |
| MASH1 R | antisense | genomic | AGCAAGAACTTTCAGCTGTGCGTG |
Statistical analysis
All quantified data were statistically analyzed and presented as mean ± SEM. Two-tailed Student’s t tests were used to calculate statistical significance with p values. A p value < 0.05 was considered statistically significant.
Supplementary Material
Video S1
Time lapse of astrocytic reprogramming to induced-neurons following CEND1 overexpression.
Video S2
Time lapse of astrocytic reprogramming to induced-neurons following CEND1 overexpression.
Video S3
Time lapse of astrocytic reprogramming to induced-neurons following CEND1 overexpression.
Acknowledgements
This work was supported by IKY-Postdocs Fellowship Grant to KA, as well as, KRIPIS-II Grant “Infectious, autoimmune and neurodegenerative diseases: study of the pathogenetic mechanisms and development of diagnostic, prognostic and therapeutic approaches” (MIS 5002486) implemented under the “Action for the Strategic Development on the Research and Technological Sector”, funded by the Operational Programme "Competitiveness, Entrepreneurship and Innovation" (NSRF 2014-2020) and co-financed by Greece and the European Union (European Regional Development Fund). We thank Dr. Evangelia Xingi in Hellenic Pasteur Institute Light Microscopy Unit (LMU) for support in live cell imaging.
Abbreviations
ASCL1, Achaete-Scute Family bHLH Transcription Factor 1 ; BDNF, Brain derived neurotrophic factor ; bFGF2, basic fibroblast growth factor 2 ; CEND1, Cell Cycle Exit And Neuronal Differentiation 1 ; EGF2, epidermal growth factor 2 ; GDNF, Glial cell derived neurotrophic factor ; cAMP, cyclic AMP ; CNS, Central Nervous System ; DCX, doublecortin ; FBS, Foetal bovine serum; ; GABA, Gamma-Aminobutyric Acid ; GAPDH, Glyceraldehyde 3-phosphate dehydrogenase ; GFAP, Glial fibrillary acidic protein ; Glast, Glutamate transporter ; Glut, Glutamate ; IGF, Insulin-like growth factor ; iNs, induced-neurons ; iPS, induced pluripotent stem cells ; NEAA, non-essential amino acids ; NEUROG2, Neurogenin- 2 ; NG2 cells, polydendrocytes ; PBS, phosphate buffer saline ; NPCs, neural progenitor cells ; NSCs, neural stem cells ; pCAG, chicken beta actin promoter ; PF, paraformaldehyde ; RV, retrovirus ; SOX2, SRY-Box Transcription Factor 2 ; SVZ, subventricular zone ; Syn1, Synapsin 1 ; TH, Tyrosine hydroxylase ; VPA, Valproic acid ;References
Aravantinou-Fatorou K., Ortega F., Chroni-Tzartou D., Antoniou N., Poulopoulou C., Politis P. K., Berninger B., Matsas R., Thomaidou D. (2015). CEND1 and NEUROGENIN2 Reprogram Mouse Astrocytes and Embryonic Fibroblasts to Induced Neural Precursors and Differentiated Neurons. Stem Cell Reports 5: 405-418.
Arlotta P., Berninger B. (2014). Brains in metamorphosis: reprogramming cell identity within the central nervous system. Current Opinion in Neurobiology 27: 208-214.
Berninger B., Costa M. R., Koch U., Schroeder T., Sutor B., Grothe B., Gotz M. (2007). Functional Properties of Neurons Derived from In Vitro Reprogrammed Postnatal Astroglia. Journal of Neuroscience 27: 8654-8664.
Biswas D., Jiang P. (2016). Chemically Induced Reprogramming of Somatic Cells to Pluripotent Stem Cells and Neural Cells. International Journal of Molecular Sciences 17: 226.
Burda J. E., Sofroniew M. V. (2014). Reactive Gliosis and the Multicellular Response to CNS Damage and Disease. Neuron 81: 229-248.
Chandrasekaran A., Avci H. X., Leist M., Kobolák J., Dinnyés A. (2016). Astrocyte Differentiation of Human Pluripotent Stem Cells: New Tools for Neurological Disorder Research. Frontiers in Cellular Neuroscience 10: 215.
Chen G., Wernig M., Berninger B., Nakafuku M., Parmar M., Zhang C.L. (2015). In Vivo Reprogramming for Brain and Spinal Cord Repair . eneuro 2: ENEURO.0106-15.2015.
Cheng L., Gao L., Guan W., Mao J., Hu W., Qiu B., Zhao J., Yu Y., Pei G. (2015). Direct conversion of astrocytes into neuronal cells by drug cocktail. Cell Research 25: 1269-1272.
Colasante G., Rubio A., Massimino L., Broccoli V. (2019). Direct Neuronal Reprogramming Reveals Unknown Functions for Known Transcription Factors. Frontiers in Neuroscience 13: 283.
Costa M. R., Ortega F., Brill M. S., Beckervordersandforth R., Petrone C., Schroeder T., Götz M., Berninger B. (2011). Continuous live imaging of adult neural stem cell division and lineage progression in vitro. Development 138: 1057-1068.
Gaitanou M., Segklia K., Matsas R. (2019). Cend1, a Story with Many Tales: From Regulation of Cell Cycle Progression/Exit of Neural Stem Cells to Brain Structure and Function. Stem Cells International 2019: 1-16.
Gao L., Guan W., Wang M., Wang H., Yu J., Liu Q., Qiu B., Yu Y., Ping Y., Bian X., Shen L., Pei G. (2017). Direct Generation of Human Neuronal Cells from Adult Astrocytes by Small Molecules. Stem Cell Reports 8: 538-547.
Gascón S., Murenu E., Masserdotti G., Ortega F., Russo G. L., Petrik D., Deshpande A., Heinrich C., Karow M., Robertson S. P., Schroeder T., Beckers J., Irmler M., Berndt C., Angeli J. P. F., Conrad M., Berninger B., Götz M. (2016). Identification and Successful Negotiation of a Metabolic Checkpoint in Direct Neuronal Reprogramming. Cell Stem Cell 18: 396-409.
Goldman S. A. (2016). Stem and Progenitor Cell-Based Therapy of the Central Nervous System: Hopes, Hype, and Wishful Thinking. Cell Stem Cell 18: 174-188.
Guo Z., Zhang L., Wu Z., Chen Y., Wang F., Chen G. (2014). In Vivo Direct Reprogramming of Reactive Glial Cells into Functional Neurons after Brain Injury and in an Alzheimer’s Disease Model. Cell Stem Cell 14: 188-202.
Haim L. B., Rowitch D. H. (2016). Functional diversity of astrocytes in neural circuit regulation. Nature Reviews Neuroscience 18: 31-41.
Han X., Chen M., Wang F., Windrem M., Wang S., Shanz S., Xu Q., Oberheim N. A., Bekar L., Betstadt S., Silva A. J., Takano T., Goldman S. A., Nedergaard M. (2013). Forebrain Engraftment by Human Glial Progenitor Cells Enhances Synaptic Plasticity and Learning in Adult Mice. Cell Stem Cell 12: 342-353.
Hashemian S., O’Rourke C., Phillips J. B., Strömberg I., af Bjerkén S. (2015). Embryonic and mature astrocytes exert different effects on neuronal growth in rat ventral mesencephalic slice cultures. SpringerPlus 4: 558.
Heinrich C., Blum R., Gascón S., Masserdotti G., Tripathi P., Sánchez R., Tiedt S., Schroeder T., Götz M., Berninger B. (2010). Directing Astroglia from the Cerebral Cortex into Subtype Specific Functional Neurons. PLoS Biology 8: e1000373.
Heinrich C., Gascón S., Masserdotti G., Lepier A., Sanchez R., Simon-Ebert T., Schroeder T., Götz M., Berninger B. (2011). Generation of subtype-specific neurons from postnatal astroglia of the mouse cerebral cortex. Nature Protocols 6: 214-228.
Hsieh J., Nakashima K., Kuwabara T., Mejia E., Gage F. H. (2004). Histone deacetylase inhibition-mediated neuronal differentiation of multipotent adult neural progenitor cells. Proceedings of the National Academy of Sciences 101: 16659-16664.
Hu W., Qiu B., Guan W., Wang Q., Wang M., Li W., Gao L., Shen L., Huang Y., Xie G., Zhao H., Jin Y., Tang B., Yu Y., Zhao J., Pei G. (2015). Direct Conversion of Normal and Alzheimer’s Disease Human Fibroblasts into Neuronal Cells by Small Molecules. Cell Stem Cell 17: 204-212.
Huang Y., Tan S. (2015). Direct lineage conversion of astrocytes to induced neural stem cells or neurons. Neuroscience Bulletin 31: 357-367.
Jung G.A., Yoon J.Y., Moon B.S., Yang D.H., Kim H.Y., Lee S.H., Bryja V., Arenas E., Choi K.Y. (2008). Valproic acid induces differentiation and inhibition of proliferation in neural progenitor cells via the beta-catenin-Ras-ERK-p21Cip/WAF1 pathway. BMC Cell Biology 9: 66.
Katsimpardi L., Gaitanou M., Malnou C. E., Lledo P.M., Charneau P., Matsas R., Thomaidou D. (2008). BM88/Cend1 Expression Levels Are Critical for Proliferation and Differentiation of Subventricular Zone-Derived Neural Precursor Cells. Stem Cells 26: 1796-1807.
Liu M.L., Zang T., Zou Y., Chang J. C., Gibson J. R., Huber K. M., Zhang C.L. (2013). Small molecules enable neurogenin 2 to efficiently convert human fibroblasts into cholinergic neurons. Nature Communications 4: 2183.
Lundin A., Delsing L., Clausen M., Ricchiuto P., Sanchez J., Sabirsh A., Ding M., Synnergren J., Zetterberg H., Brolén G., Hicks R., Herland A., Falk A. (2018). Human iPS-Derived Astroglia from a Stable Neural Precursor State Show Improved Functionality Compared with Conventional Astrocytic Models. Stem Cell Reports 10: 1030-1045.
Magnusson J. P., Zamboni M., Santopolo G., Mold J. E., Barrientos-Somarribas M., Talavera-Lopez C., Andersson B., Frisén J. (2020). Activation of a neural stem cell transcriptional program in parenchymal astrocytes. eLife 9: e59733.
Makri G., Lavdas A. A., Katsimpardi L., Charneau P., Thomaidou D., Matsas R. (2010). Transplantation of Embryonic Neural Stem/Precursor Cells Overexpressing BM88/Cend1 Enhances the Generation of Neuronal Cells in the Injured Mouse Cortex . Stem Cells 28: 127-139.
Mertens J., Paquola A. C.M., Ku M., Hatch E., Böhnke L., Ladjevardi S., McGrath S., Campbell B., Lee H., Herdy J. R., Gonçalves J. T., Toda T., Kim Y., Winkler J., Yao J., Hetzer M. W., Gage F. H. (2015). Directly Reprogrammed Human Neurons Retain Aging-Associated Transcriptomic Signatures and Reveal Age-Related Nucleocytoplasmic Defects. Cell Stem Cell 17: 705-718.
Niu W., Zang T., Zou Y., Fang S., Smith D. K., Bachoo R., Zhang C.L. (2013). In vivo reprogramming of astrocytes to neuroblasts in the adult brain. Nature Cell Biology 15: 1164-1175.
Oberheim N. A., Goldman S. A., Nedergaard M. (2012). Heterogeneity of Astrocytic Form and Function. In Astrocytes. (Ed. Milner Richard) Humana Press, Totowa, NJ.
Ortega F., Costa M. R., Simon-Ebert T., Schroeder T., Götz M., Berninger B. (2011). Using an adherent cell culture of the mouse subependymal zone to study the behavior of adult neural stem cells on a single-cell level. Nature Protocols 6: 1847-1859.
Papadodima O., Sergaki M., Hurel C., Mamalaki A., Matsas R. (2005). Characterization of the BM88 promoter and identification of an 88 bp fragment sufficient to drive neurone-specific expression. Journal of Neurochemistry 95: 146-159.
Péron S., Berninger B. (2015). Reawakening the sleeping beauty in the adult brain: neurogenesis from parenchymal glia. Current Opinion in Genetics & Development 34: 46-53.
Politis P. K., Makri G., Thomaidou D., Geissen M., Rohrer H., Matsas R. (2007). BM88/CEND1 coordinates cell cycle exit and differentiation of neuronal precursors. Proceedings of the National Academy of Sciences 104: 17861-17866.
Robertson J. M. (2014). Astrocytes and the evolution of the human brain. Medical Hypotheses 82: 236-239.
Rusnakova V., Honsa P., Dzamba D., Ståhlberg A., Kubista M., Anderova M. (2013). Heterogeneity of Astrocytes: From Development to Injury – Single Cell Gene Expression. PLoS ONE 8: e69734.
Smith D. K., Yang J., Liu M.L., Zhang C.L. (2016). Small Molecules Modulate Chromatin Accessibility to Promote NEUROG2-Mediated Fibroblast-to-Neuron Reprogramming. Stem Cell Reports 7: 955-969.
Su Z., Niu W., Liu M.L., Zou Y., Zhang C.L. (2014). In vivo conversion of astrocytes to neurons in the injured adult spinal cord. Nature Communications 5: 3338.
Takahashi K., Yamanaka S. (2006). Induction of Pluripotent Stem Cells from Mouse Embryonic and Adult Fibroblast Cultures by Defined Factors. Cell 126: 663-676.
Torper O., Pfisterer U., Wolf D. A., Pereira M., Lau S., Jakobsson J., Björklund A., Grealish S., Parmar M. (2013). Generation of induced neurons via direct conversion in vivo. Proceedings of the National Academy of Sciences 110: 7038-7043.
Wu X., Liu Y., Chen X., Sun Q., Tang R., Wang W., Yu Z., Xie M. (2013). Involvement of TREK-1 Activity in Astrocyte Function and Neuroprotection Under Simulated Ischemia Conditions. Journal of Molecular Neuroscience 49: 499-506.
Zhang L., Yin J.C., Yeh H., Ma N.X., Lee G., Chen X. A., Wang Y., Lin L., Chen L., Jin P., Wu G.Y., Chen G. (2015). Small Molecules Efficiently Reprogram Human Astroglial Cells into Functional Neurons. Cell Stem Cell 17: 735-747.
Zheng W., Li Q., Zhao C., Da Y., Zhang H.L., Chen Z. (2018). Differentiation of Glial Cells From hiPSCs: Potential Applications in Neurological Diseases and Cell Replacement Therapy. Frontiers in Cellular Neuroscience 12: 239.